Drug Development Readiness: Interactive Checklist and RACI
Align early, succeed faster: A RACI-based guide to multi-disciplinary drug development
Drug development is a complex, multi-disciplinary endeavor. Scientific excellence is at the basis of its success. However, if not complemented by timely and coordinated input from a broad range of expertise spanning various disciplines, it won’t be sufficient.
This raises a critical question: which expertise is needed, at what point in development, and to provide which inputs in order to build a comprehensive and robust drug development plan?
Align your team, share a common vision, and keep every discipline informed and inspired from day one to drive program success
RACI Matrix
The RACI matrix below is designed to help address this question by mapping required disciplines and their key contributions, enabling structured, cross-functional planning from the start.
Early decisions can have long-lasting consequences for development timelines, cost, risk, and overall probability of success. Engaging the appropriate disciplines early helps avoid downstream surprises and rework.
Drug Development RACI Matrix
Responsibility assignment matrix for drug development programs
| Activity / Objective | Discovery/Research | Molecular biology/ protein engineering | Immunology/ pharmacology | Translational medicine | Bioinformatics/ computational biology | CMC/ Manufacturing | Toxicology/ Safety | Regulatory affairs | Clinical development/ medical affairs | Project management/ QC | Quality assured/ QC | IP counsel |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Target identification and validation | R | C | C | C | C | I | I | C | I | A | I | C |
| Initial antibody generation / screening | R | C | C | C | C | I | I | C | I | A | I | C |
| Epitope mapping and specificity testing | C | R | C | C | R | I | I | C | I | A | I | C |
| Lead antibody selection | R | R | R | R | C | C | C | C | I | A | I | C |
| Antibody engineering and humanization | C | R | C | C | C | I | I | C | I | A | I | C |
| Fc / functional optimization | C | R | C | C | C | I | I | C | I | A | I | C |
| In vitro functional assays | C | C | R | C | C | I | C | C | I | A | I | C |
| In vivo efficacy / PK-PD studies | C | C | R | C | C | C | C | C | I | A | I | C |
| Bioinformatics off-target / immunogenicity prediction | C | C | C | C | R | I | I | C | I | A | I | C |
| Manufacturing feasibility | C | C | I | I | C | R | I | C | I | A | C | C |
| Preclinical candidate selection | R | R | R | R | C | C | C | C | I | A | I | C |
| Formulation and stability | I | C | I | C | I | R | I | C | I | A | C | I |
| Analytical method development | I | C | I | C | I | R | I | C | I | A | C | I |
| CMC / GMP batch development | I | C | I | C | I | R | I | C | I | A | C | I |
| GLP toxicology studies | I | I | C | C | C | C | R | C | I | A | C | I |
| Safety pharmacology / immunogenicity | I | I | C | C | C | C | R | C | I | A | C | I |
| IND-enabling studies completion | I | I | C | C | C | C | R | C | I | A | C | I |
| Regulatory strategy and IND submission | I | I | I | C | I | C | C | R | C | A | C | I |
| Phase I trial design and biomarker strategy | I | I | C | R | C | I | C | C | R | A | I | I |
| Clinical site selection and trial initiation | I | I | I | C | I | I | I | C | R | A | I | I |
| First patient dosed | I | I | I | C | I | I | C | C | R | A | I | I |
Early decisions can have long-lasting consequences for development timelines, cost, risk, and overall probability of success. Engaging the appropriate disciplines early helps avoid downstream surprises and rework.
Readiness Checklist
Use this interactive checklist to evaluate and strengthen the multidisciplinary foundation of your drug development plan. It is intended to:
- Clarify which areas of expertise are needed
- Highlight what each discipline should contribute to the drug development plan
- Identify gaps, delays, or misalignment in cross-functional involvement
- Encourage integrated planning rather than siloed decision-making
By identifying missing expertise early, you can close gaps proactively and avoid downstream impacts on critical decisions, timelines, and program success. Developed with antibody programs as a reference, it is a practical, adaptable tool to guide cross-functional planning from early discovery through late-stage development.
Drug Development Expertise Readiness Checklist
Confirm that all critical capabilities across the development lifecycle are covered and clearly owned.
| Capability / status and responsibility | Status | In House / Outsourced / Mixed | Owner/Vendor | |
|---|---|---|---|---|
| 1 | Discovery & Translational Science | |||
| Target identification and validation | ||||
| Antibody engineering / optimization | ||||
| In vitro / in vivo pharmacology | ||||
| Biomarker strategy | ||||
| Translational science alignment | ||||
| Immunogenicity | ||||
| 2 | Preclinical and CMC | |||
| Manufacturability | ||||
| Cell line development | ||||
| Process development (USP/DSP) | ||||
| Analytical development | ||||
| Formulation and stability | ||||
| GMP manufacturing | ||||
| GLP toxicology and safety pharmacology | ||||
| IND-enabling strategy | ||||
| 3 | Clinical Development | |||
| Clinical trial design | ||||
| Regulatory submissions (IND/CTA) | ||||
| Clinical operations | ||||
| Biostatistics & data management | ||||
| Pharmacovigilance | ||||
| Regulatory authority interactions | ||||
| 4 | Quality and Manufacturing Oversight | |||
| Quality assurance (QA) | ||||
| Quality control (QC) | ||||
| Tech transfer management | ||||
| Supply chain management | ||||
| 5 | Strategic and Business Capabilities | |||
| Regulatory strategy (long-term) | ||||
| IP ownership & lifecycle management | ||||
| Competitive landscape analysis | ||||
| Partnering / licensing readiness | ||||
| Financial modeling and valuation | ||||
| Program / project management | ||||
It is important to note that the project manager is accountable at a project coordination level, ensuring milestones are met and activities are aligned across functions, and communication flows effectively between teams. For each technical deliverable, however, the accountability lies with the functional leader (scientific, clinical, regulatory or CMC) who has decision-making authority within their particular domain.
For major program decisions, particularly the selection of the final clinical candidate, governance typically extends beyond the functional lead. In many organizations, the head of discovery proposes a candidate to senior management and scientific board, who review the data package and formally approve the selection of the clinical candidate.
In addition, intellectual property protection is a critical activity that runs in parallel with scientific development. Securing IP generally requires coordination between Research/Discovery, Business Development and IP Counsel, ensuring that patent filings appropriately protect the therapeutic candidate and its potential commercial value. 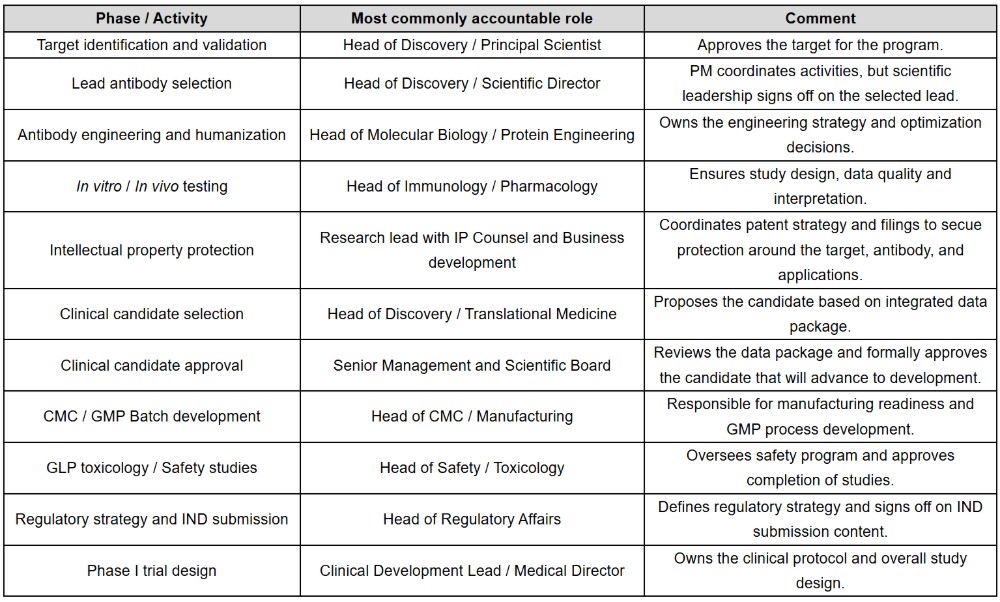
These resources were developed in collaboration with Marta Kijanka, PhD, a medical biotechnologist and founder of MK Bio Consultancy.
At CDMO Live Europe 2026, Marta will be hosting the Biotech CMC Bootcamp, designed to explore the CMC strategies that separate funded biotechs from failed ones.

Marta advises CDMOs and biotech companies on CMC, project management, vendor selection, technical transfers, and process scale-ups, and provides tailored industry training. Based in Oss, The Netherlands, her services include comprehensive operational assessments of CDMOs — going beyond GMP audits to evaluate organizational maturity, project management capabilities, capacity planning, and strategic fit, helping clients make informed partnering decisions and flag execution risks early.

