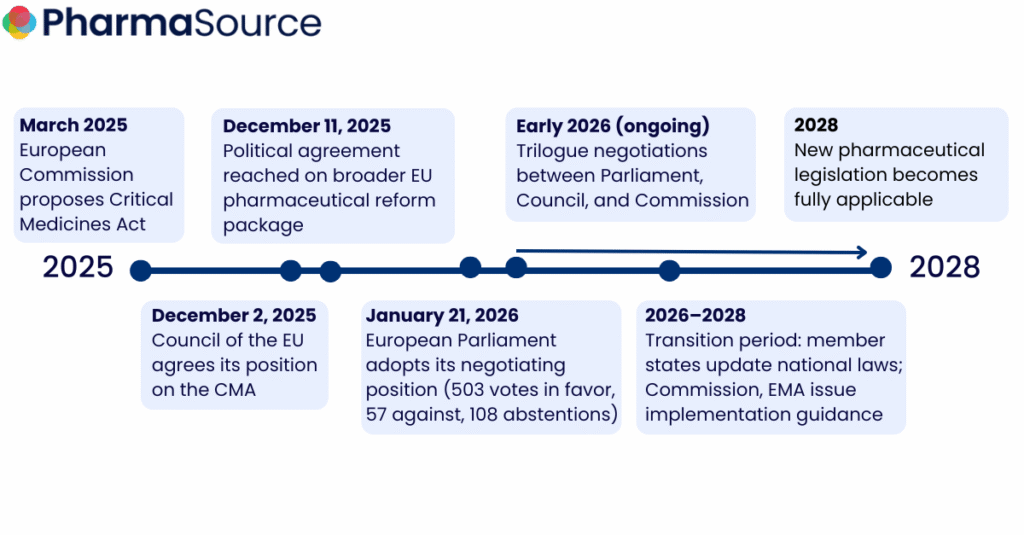
Europe took a decisive step toward pharmaceutical sovereignty on January 21, when the European Parliament overwhelmingly approved the Critical Medicines Act. With 503 MEPs backing the proposal out of 668, the legislation now enters trilogue negotiations (three-way talks between Parliament, Council, and Commission) that will determine how pharmaceutical companies must restructure their European manufacturing strategies over the next decade.
From Price to Resilience
Under the Parliament’s negotiating position, EU contracting authorities would be required to favor producers manufacturing at least 50% of critical medicines or active pharmaceutical ingredients within the EU.
Current procurement practices award contracts primarily based on the lowest price, driving production to Asian manufacturers. The proposed framework mandates that security of supply and manufacturing location take precedence over cost considerations.
“Price can no longer be the sole criterion when awarding contracts for medicines,” said rapporteur Tomislav Sokol (EPP, Croatia) following the vote. “Security of supply, the resilience of production chains, and manufacturing capacity within the EU must be taken into account.”
This would mean that companies relying on the lowest-cost Asian API suppliers for critical medicines face a competitive disadvantage in EU public procurement if these provisions survive trilogue negotiations. Those with established or planned EU manufacturing capacity would gain preferential treatment.

Manufacturing Strategy Implications
Three elements of the Parliament’s position create specific planning imperatives for external manufacturing leaders:
Strategic Projects designation. The Act establishes a framework for industrial projects that create, modernize, or expand EU manufacturing capacity for critical medicines. Designated projects would receive fast-tracked permits, regulatory support, and priority access to EU and national funding.
For companies planning European capacity investments, timing matters. Projects positioned for Strategic Project designation could access significantly reduced approval timelines and state aid that competitors without this status cannot access.
Collaborative procurement threshold reduction. The Parliament proposes lowering the number of member states required to trigger joint procurement from nine to five, while the Council agreed in December 2025 to reduce the threshold to six. This divergence will be negotiated during trilogue, but either outcome makes cross-border purchasing agreements more accessible, particularly for smaller countries that previously lacked negotiating leverage.
These changes suggest that external manufacturing teams will likely face increased requests for proposals from multi-country consortia, with evaluation criteria weighted toward supply security rather than unit price. RFP response strategies built around the lowest cost bids will require recalibration.
Stockpile coordination and redistribution. The most contentious element heading into trilogue negotiations is the Parliament’s proposal to empower the European Commission to mandate the redistribution of medicines from one member state’s stockpile to another during critical shortages.
“What we have now, unfortunately, is that national stockpiling in some member states, especially the big ones, directly contributes to shortages in small member states,” Sokol explained during the post-vote press conference.
This redistribution mechanism, if adopted, would create supply planning complexity for manufacturers. Contracts tied to national stockpiles may face unexpected allocation changes during shortage events, requiring flexible inventory management and transparent visibility across the European network.
The Cost Trade-Off
Industry estimates suggest the Act could raise prices for critical medicines by 20–40%. Several factors contribute to this increase. Namely, labor and facility costs in the EU are higher than in Asia, diversified supply chains require extra production capacity, and mandatory stockpiles tie up significant working capital.
Industry Pushback Centers on Procurement Flexibility
The European Federation of Pharmaceutical Industries and Associations (EFPIA) supports supply resilience objectives but has raised specific concerns about execution. Director General Nathalie Moll emphasized the need for the Act to “focus on real supply risks and practical solutions.”
EFPIA’s primary concerns center on procurement requirements. The organization argues that favoring manufacturing location “risks weakening global supply diversification, reducing flexibility in supply chains and undermining Europe’s position as an export-oriented base for innovative medicines.”
The pharma industry has also been pushing back against the lowered threshold for joint procurement, warning these mechanisms could dilute the Act’s original intent.
The Parliament included safeguards such as binding quantity commitments to address industry concerns, but differing priorities remain. European health authorities want security of supply regardless of cost, while industry seeks profitable pricing that justifies European manufacturing investment.
Global Supply Chain Concerns
The Act’s stockpiling requirements have drawn criticism from global health organizations concerned about unintended consequences in developing markets. Amref Health Africa warns of three risks:
- EU stockpile mandates could drain global markets of limited API supplies
- Increased EU demand could drive up commodity prices, making medicines unaffordable in lower-income markets
- “Buy European” incentives may undercut pharmaceutical manufacturing development in Africa
Boniface Mbuthia, Amref’s director of health financing, warned that “without coordination, EU stockpiling could lead to supply diversion, reducing availability of medicines in African markets.”
Social Democratic MEP Wölken acknowledged these concerns: “We must ensure that European resilience is not bought at the cost of security of supply in the Global South.” However, he admitted the Act’s mechanisms focus primarily on internal EU considerations rather than external implications.
The Parliament’s focus during negotiations prioritized EU candidate countries, such as those in the Western Balkans and Ukraine, rather than partners in Africa, according to Sokol’s comments at the post-vote press conference.
What Comes Next
With the Parliament’s position now adopted, trilogue negotiations between Parliament, Council, and Commission will determine the final legislative text. Sokol expressed hope for agreement “within a couple of months,” citing urgency around US protectionist trade trends.
Key negotiation points to watch:
- Stockpile redistribution authority: The Council is expected to resist Commission power to mandate medicine transfers between member states, viewing health as a sovereign competence
- Procurement local content thresholds: The 50% EU manufacturing requirement may face adjustment based on industry feedback and supply chain realities
- Joint procurement thresholds: Whether the Parliament’s five-member-state minimum or the Council’s six-state threshold prevails
- Strategic Project eligibility criteria: Definition of which projects qualify for fast-track treatment and state aid access


 Sign up to the free weekly newsletter for all the latest news, resources and podcasts in pharma manufacturing.
Sign up to the free weekly newsletter for all the latest news, resources and podcasts in pharma manufacturing.