- AsymBio, a subsidiary of Asymchem Group, has launched its Fengxian commercial manufacturing site in Shanghai, completing a three-site CDMO network in the city.
- The Shanghai network integrates early research, clinical pilot production, and commercial manufacturing to support biologics programs from IND to BLA.
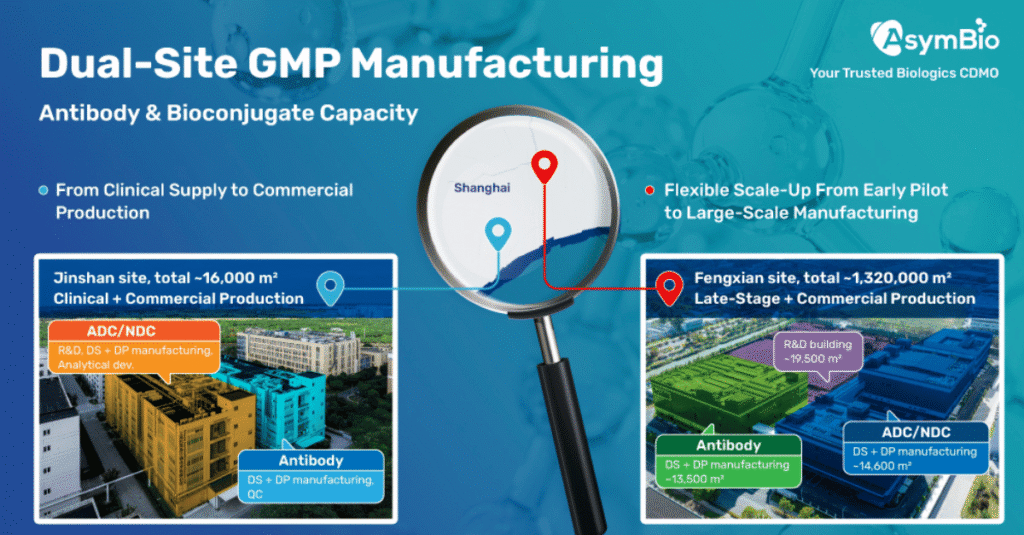
AsymBio, a subsidiary of Asymchem Group, has commenced operations at its Fengxian commercial manufacturing site in Shanghai. The launch completes the company’s Shanghai-based “Golden Triangle” network, which also includes the Zhangjiang Science & Technology Center and the Jinshan manufacturing site.
The three locations form an integrated contract development and manufacturing organization (CDMO) platform designed to support biologics programs across the development lifecycle. According to the company, the network connects early research and process development with clinical pilot-scale manufacturing and commercial production to help clients advance programs from IND to BLA.
The Zhangjiang Science & Technology Center focuses on early-stage research and technology development, including cell line development, upstream and downstream process development, conjugation technologies, and analytical method development. The Jinshan site provides manufacturing services ranging from clinical supply to commercial production for antibodies, protein therapeutics, antibody-drug conjugates (ADCs), and novel drug conjugates (NDCs).
The newly commissioned Fengxian facility is designed for late-stage development and large-scale commercial supply of biologics. It incorporates smart manufacturing systems and GMP infrastructure, including single-use bioreactors for antibody drug substance production ranging from 200 L to 2,000 L, conjugation reactors between 50 L and 500 L, and automated filling and lyophilization systems supporting multiple vial formats.
AsymBio also connects its Shanghai manufacturing operations with Asymchem Group’s payload–linker research and manufacturing facility in Tianjin. This structure links payload and linker development with antibody drug substance, conjugate drug substance, and drug product manufacturing, enabling integrated contract manufacturing services for biologics and drug conjugates from early development through commercialization.

















