“The wake-up call happened during COVID — we realized our dependency on basic medicines coming from outside Europe was dangerously high.”
Elisabeth Stampa, CEO of Medichem and Vice President of Medicines for Europe, has been one of the pharmaceutical industry’s most outspoken advocates for the Critical Medicines Act (CMA). With the European Parliament having just published its position and trilogue negotiations underway, she argues the window for meaningful reform is open — but not indefinitely.
In the latest PharmaSource podcast episode, Elisabeth breaks down what the CMA is designed to achieve, where the friction points lie, and what success would actually look like for European patients and the generics and biosimilar industry by 2030.
The Origins of the Critical Medicines Act
The CMA didn’t emerge from a policy vacuum. COVID-19 exposed just how dependent Europe had become on medicines — primarily older, off-patent molecules used in emergency settings — sourced overwhelmingly from outside the continent. When borders closed, supply chains buckled.
“Immediately after COVID, when we started to go back to normal life, the Russia-Ukraine conflict started,” Elisabeth notes. “That got a lot of attention from European authorities.”
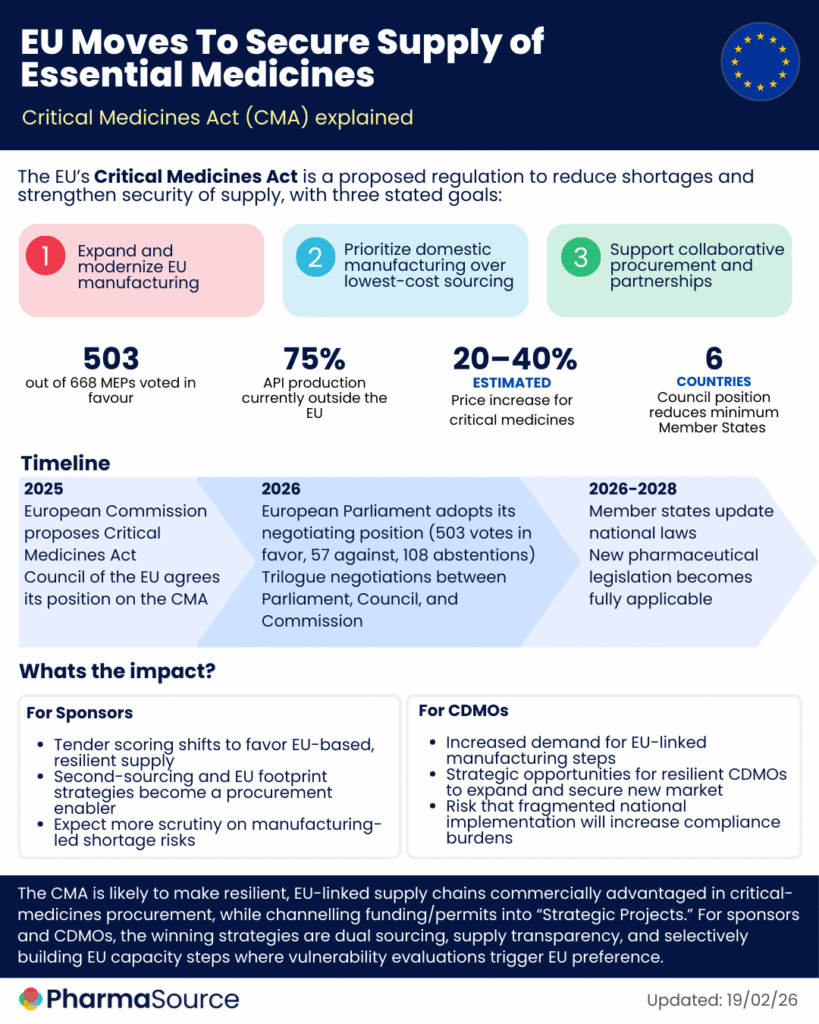
The Belgian Presidency of the European Council drove the initial push for a Critical Medicines Alliance, which evolved into the legislative proposal now known as the CMA. The effort was coordinated through HERA — the EU’s Health Emergency Preparedness and Response Authority — and brought together an unusually broad coalition: patient associations, hospitals, pharmacists, regulators, and both innovators and generic and biosimilar industry representatives.
The CMA centers on a list of roughly 300 critical molecules, updated annually. According to IQVIA, over 90% are generics — older molecules, many of which are no longer manufactured in Europe at all.
What the CMA Is Actually Trying to Do
The legislation targets three interconnected problems: supply chain dependency, chronic medicine shortages, and a procurement system that has driven prices to unsustainable lows.
“It’s trying to reduce dependency on a certain group of medicines and strengthen the industry in Europe,” Elisabeth explains. “But it’s also trying to regulate the root causes of shortages — and the main root cause is downward price pressure and tenders based only on price.”
The Parliament’s version of the CMA goes further than the Commission’s original text. It introduces provisions on stockpiling — addressing the imbalance created when larger member states maintain reserves that effectively deprive neighboring countries of supply. It also pushes for sourcing preferences toward products manufactured wholly or partly in the EU.
On collaborative procurement, Elisabeth draws an important distinction. For rare and orphan drugs — small volumes, complex supply — joint procurement across member states makes sense. For high-volume generics, it’s far more complicated: 27 member states, multiple packaging formats, at least 15 languages, and separate national licensing rules.
The Pricing Problem No Policy Can Easily Fix
Perhaps the most sobering part of the conversation is Elisabeth’s data point on generics pricing: over the past five years, the price of bread has risen 40%. The price of generics has fallen 8%.
“The workforce expects annual salary increases aligned with inflation. Energy costs go up. Everything goes up. Pricing should at least be indexed to inflation — and it isn’t.”
Medicines for Europe is advocating for the Most Economically Advantageous Tender (MEAT) criteria — a framework that weighs factors beyond price, including supply resilience and the ability to dual-source from multiple awarded suppliers. The problem, Elisabeth is frank about, is member states have they own health budgets, driven by their national priorities.
“Not all the members agree or can afford to pay a higher price for products manufactured in Europe.”
That tension — between European-level ambition and national-level fiscal autonomy — is potentially the most significant blocker to ratification.
The US Parallel: Reshoring Has Its Own Limits
Elisabeth draws a direct comparison with US reshoring efforts, including advanced manufacturing investments in continuous flow technologies and federal funding for domestic API production. Her assessment is measured.
“Continuous flow for API production is only applicable to certain molecules — it’s not one solution that works for all.” Labor costs in the US are among the highest globally, which may undermine the economic case for large-scale domestic production. And critically, for many APIs, the key starting materials are sourced 100% from Southeast Asia — and in some cases the key starting material cannot be produced in Europe due to environmental regulations.
“Regulators know which API are registered and approved for each finished product, but don’t have updated and complete information which API is used in every single production — neither in the US nor in Europe.”
Her broader point: neither Europe nor the US will be able to manufacture 100% of their essential medicines domestically, nor should they try. The more realistic objective is building resilience through allied-country sourcing — treating European and US manufacturers as strategic partners rather than competitors.
“Relying on Europe would make the US less dependent on other territories. And the same works in reverse.”
What Success Looks Like by 2030
The CMA is expected to complete trilogue negotiations through 2026. That gives member states roughly two years to transpose the legislation — making measurable impact assessable by around 2030.
Elisabeth offers three indicators of success: whether specific critical molecules have been effectively reshored to Europe; whether generic pricing has stabilized rather than continued its downward trend; and — most fundamentally — whether the frequency of medicine shortages has declined.
“If the number of shortages has been reduced, that’s the main measure. That’s the whole point.”
On the broader opportunity, she is candid about her frustration with Europe’s pace. “I feel a bit sad when I hear how much other territories — India, China, the US — are advancing. I think we are missing our opportunity. Establishing the Critical Medicines Alliance would be extremely good for European patients and for the European industry.”











