Contract development and manufacturing invested a disclosed amount of $7.2 billion in September 2025—with 97% flowing to US facilities—as the sector’s reshoring momentum reached the highest level of the year so far.
PharmaSource tracks every significant CDMO announcement globally—partnerships, investments, facility expansions, executive moves, and more. Our free CDMO News Tracker, updated daily, gives you real-time visibility into what’s happening across contract manufacturing. Here’s what September’s data revealed.
Activity Surges: Highest Monthly Volume of 2025
September recorded 81 announcements—up 55% on August and the highest monthly total this year. Nearly half (39 announcements) were partnerships, whilst 18 new facility expansions were announced.
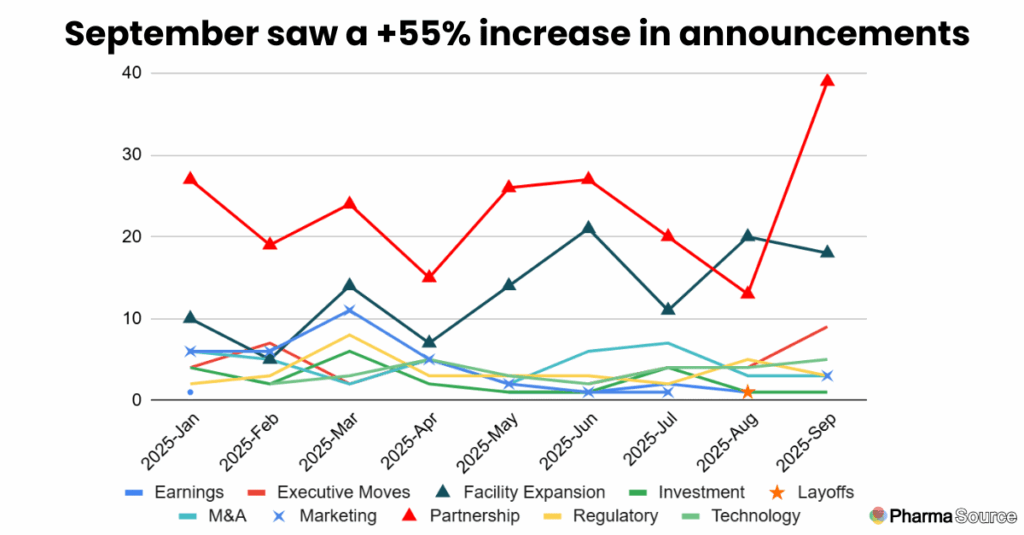
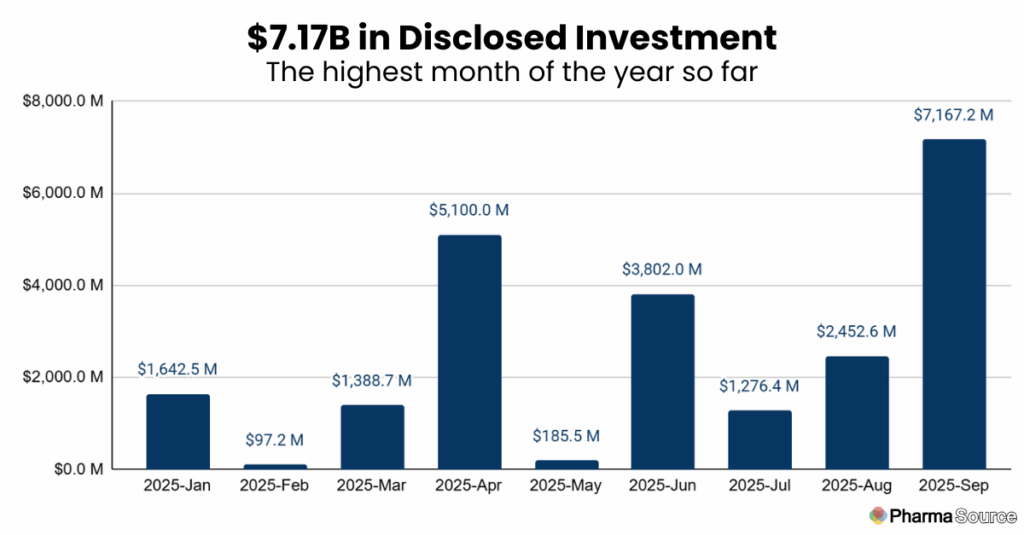
Investment Highlights: Record $7.2 Billion Month
September delivered the highest disclosed investment volume of 2025 at $7.17 billion—nearly triple August’s $2.45 billion and surpassing April’s previous high of $5.1 billion.
Remarkably, 97% ($6.7 billion) of disclosed investment targeted the United States, led by:
- FUJIFILM Biotechnologies opened its $3.2 billion Holly Springs biomanufacturing facility—the single largest investment of the month, underscoring the industry’s bullish outlook on biologics manufacturing capacity
- Hans Scientific announced an ambitious $2 billion investment to expand US drug manufacturing operations
- Samsung Biologics secured a $1.3 billion contract with a US pharmaceutical partner, marking one of the largest CDMO deals announced this year
European activity remained robust with PolyPeptide Group’s €100 million CDMO project in Malmö, AGC Pharma Chemicals’ €110 million Barcelona plant expansion, and Afton Scientific’s $200 million Virginia expansion. UK developments included Kindeva’s 150,000 square foot headquarters in Loughborough and Almac Group’s multi-million-pound investment in bioavailability technologies.
Year to date, nearly three quarters (73%) of disclosed investment is being directed to the United States.
Geographic Distribution: US Reshoring Accelerates
United States-based announcements dominated at 37% of all geographically-specific news—reinforcing continued reshoring momentum. The United Kingdom held second position at approximately 10%, followed by India (8%), and Switzerland, Italy, and Spain each contributing around 5% of activity.
Asian markets including South Korea, Thailand, and Taiwan featured prominently in facility expansion and partnership announcements, suggesting continued regional capacity buildout.
Biologics and Advanced Therapies Drive Activity
When modality was specified, biologics-related announcements represented roughly 42% of the total, reflecting sustained sector investment in large molecule manufacturing capabilities. Cell and gene therapy accounted for approximately 18% of specified announcements—a higher proportion than recent months—while small molecule manufacturing comprised around 22%.
Technology and Platform Innovation
CDMOs continued investing in proprietary technologies to differentiate their offerings:
- WuXi Biologics launched its TrueSite TI™ Platform to accelerate biologics development
- Abzena expanded its AbZelectPRO™ Platform with next-generation GS knockout systems
- Cytovance Biologics launched in-house formulation development services
- Charles River incorporated Akadeum’s GMP T Cell Isolation Kit into its Flex platform
- Integrated DNA Technologies partnered with Aldevron to launch Alt-R HDR Enhancer Protein for genome editing
Notable Executive Movement
Several high-profile CEO appointments marked September, including Kenneth Bilenberg at Avid Bioservices, Dr. Nicola Ottolina at Lisapharma, Denis Johnson at Grand River Aseptic Manufacturing, and Dr. Riku Rautsola at FAMAR. Merck appointed Kai Beckmann as next CEO effective May 2026, whilst Lonza named Jason Berndt as EVP and Head of Group Operations.
Advanced Therapy Focus
Multiple cell and gene therapy partnerships emerged, including eXmoor Pharma with Anthony Nolan CT&LS, Made Scientific with Hemogenyx for CAR-T manufacturing, and Matica Biotechnology with Cirsium Biosciences for plant-based AAV manufacturing. ProBioGen also announced it will operate a 4,600 sqm GMP facility at Berlin Center for Gene and Cell Therapies.
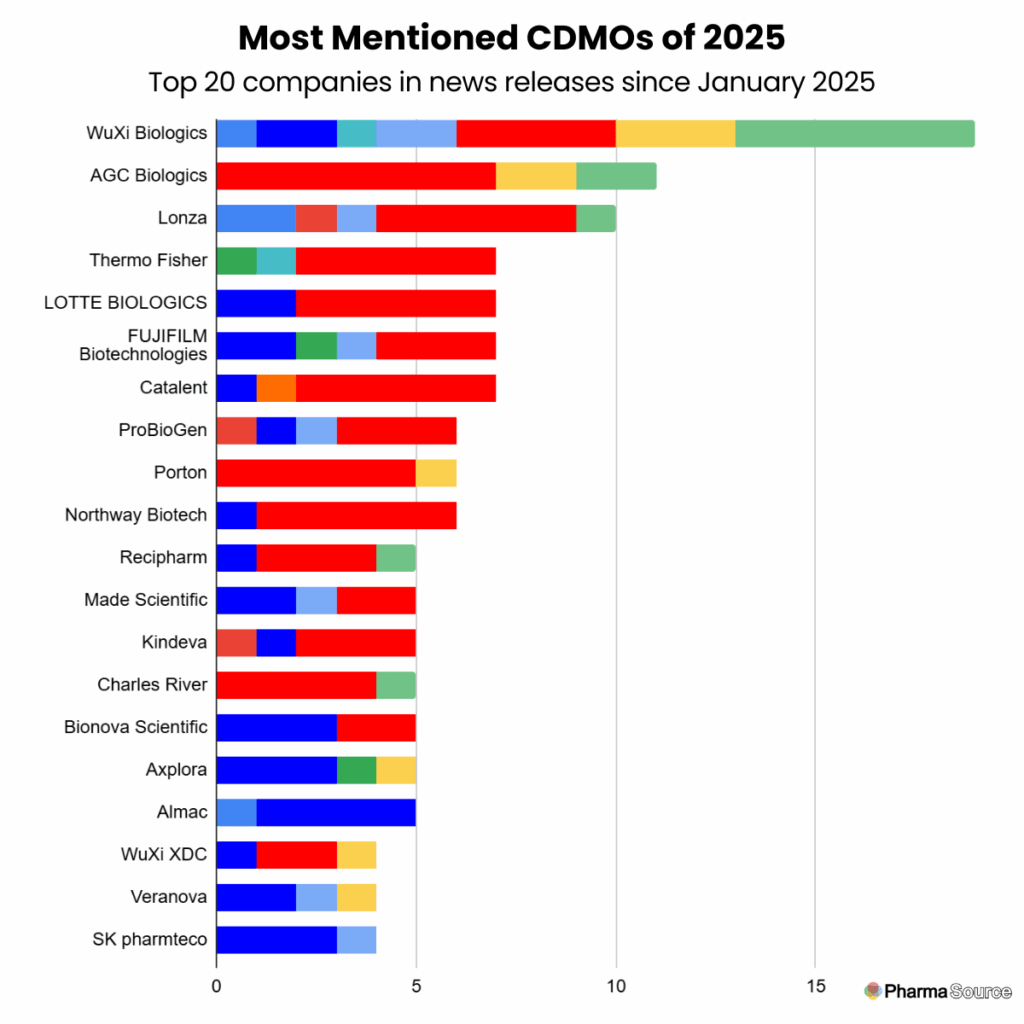
Year-to-Date: The Most Newsworthy CDMOs of 2025
Which CDMOs have been making the most noise this year? Here are the top five:
1. WuXi Biologics – 19 announcements
Leading the pack with partnerships, technology launches, and sustainability initiatives. The high activity level reflects efforts to maintain momentum following Biosecure Act disruption.
2. AGC Biologics – 11 announcements
Consistent presence across facility expansions, partnerships, and regulatory milestones.
3. Lonza – 10 announcements
A big year for one of the biggest CDMOs in the world. Notable for Chief Executive appointment and strategic partnerships, including refocus on biologics with the “One Lonza” strategy.
4. Thermo Fisher – 7 announcements
Strong activity in facility investments and strategic acquisitions.
5. LOTTE BIOLOGICS – 7 announcements
Significant facility development news, including the Incheon Bio Campus topping-out ceremony.
Other consistently active organizations include FUJIFILM Biotechnologies, Catalent, ProBioGen, and Charles River—each maintaining regular market presence through technology partnerships, executive appointments, and facility developments.
PharmaSource tracks CDMO sector announcements daily. Access the full dataset.















