“A 10% success rate means you have to be smart about every decision you make in the pipeline.”
Nick Wigdahl, Director of PharmaGro, has spent his career navigating two worlds that rarely talk to each other: the operational complexity of big pharma and the portfolio discipline of family office investing. That combination, he argues, is exactly what mid-size life science companies need more of.
Nick Wigdahl is Director of PharmaGro, a consultancy specializing in CMC, supply chain, market assessment, and portfolio management for mid-size pharmaceutical and CDMO companies. His background includes supply chain leadership roles at Roche and GSK, as well as investment and portfolio management experience with family offices in logistics and transportation.
In the latest PharmaSource podcast episode, Nick explains why managing molecule uncertainty is one of the most underappreciated disciplines in pharma, and how companies that get it right make better investment decisions, optimize their portfolios, and build more resilient businesses.
Understanding Molecule Uncertainty
Molecule uncertainty has two distinct dimensions, Nick explains: commercial uncertainty and technical or scientific uncertainty. The technical side is where most pharma companies feel the most pain.
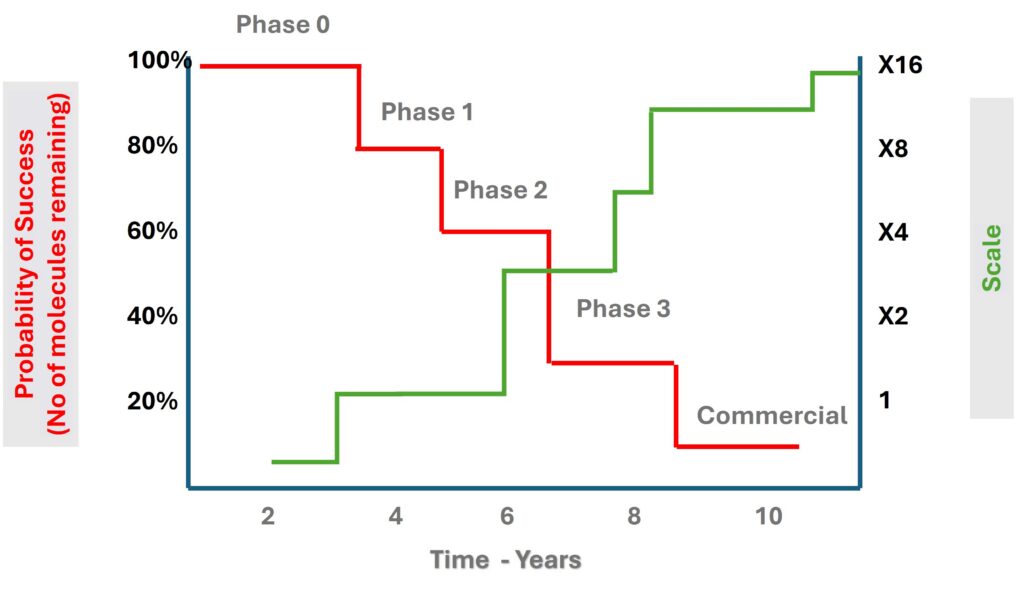
“We never know whether a molecule will succeed at each of the different phases,” he says. “There’s about a 10% chance of getting all the way from preclinical through to a commercial product.”
That 10% figure isn’t evenly distributed across the pipeline. Phase II carries the highest attrition, with success rates dropping sharply compared to Phase I, where chances sit around 50 to 66%. By Phase III, the probability recovers to roughly 70%. The preclinical stage remains the hardest to benchmark, largely because pharma companies publish far less data at that stage.
“Once you’re into the development phases, there’s a lot more spotlighting. The FDA process requires much more transparency. Preclinical is much harder to estimate.”
Understanding where risk concentrates allows companies to make smarter decisions about where to invest resources and where to pursue business development opportunities.
How AI Is Changing Pipeline Benchmarking
Historically, benchmarking phase transition probabilities required large teams of analysts trawling through FDA records. A landmark MIT study published just before 2020 set the standard for this kind of analysis. That process, Nick says, has been fundamentally transformed.
“With AI and bigger data, it’s now possible to analyze a far greater set of data, much quicker. You can start to formulate risk profiles for newer modalities, like antibody drug conjugates, in a fraction of the time it would have taken five years ago.”
Modality and therapeutic category both matter when building a risk profile. Oncology and CNS are among the highest-risk areas, partly due to longer trial durations and less well-understood modes of action. The blood-brain barrier remains a particular challenge in CNS development. Vaccines, by contrast, carry lower risk due to faster development timelines.
“Alzheimer’s and brain disease have seen some spectacular failures. The biology is more tricky, the trials are longer, and it’s harder to be sure of results.”
The ability to cut risk profiles by modality and indication gives companies a more granular and actionable view of their pipeline.
Portfolio Optimization for Mid-Size Companies
Mid-size pharma and CDMO businesses face a structural challenge: they often lack the portfolio depth to absorb the natural attrition of drug development. Nick’s work at PharmaGro focuses on helping these companies apply the kind of portfolio management discipline more commonly found in financial investment.
“You need to manage risk at each distinct phase. Understanding the potential risk helps you make better decisions around investment and resource allocation for business development.”
For CDMOs specifically, capacity and capability are two separate variables that are often conflated. Nick points to medical devices as a useful illustration.
“About 50% of products coming down pipelines now have some sort of device connected with them, and devices are getting more complicated. To train people to assemble medical devices without fault takes a lot longer than just opening a new plant. It’s a combination of capacity and capability.”
This distinction matters when evaluating CDMO partners or assessing whether internal manufacturing can support a growing portfolio. The gap between customer framing and CDMO interpretation is one of the core challenges in pipeline-stage business development.

Reshoring, Geopolitics, and the Workforce Question
The reshoring push in the US has dominated industry conversation, but Nick cautions against assuming capital is the primary constraint.
“Any shortages won’t necessarily be in areas like capital. The shortages may be in more unusual areas, like whether we have enough skilled workforce, and whether we have enough construction people to actually build the factories.”
At Roche, Nick saw this play out directly, with shortages in electricians, mechanics, and plant operators creating real bottlenecks. Analytical capability is equally underestimated.
“It’s often those sorts of roles where you can see a shortage, and analytical capability is as much a bottleneck as sheer capacity.”
Long-Term Thinking and Scenario Planning
Pharma’s product lifecycles run roughly 15 years, which creates a planning horizon that most operational teams are poorly equipped to think across. Nick’s advice is to resist the pull of the near-term and work backward from end-of-lifecycle conditions.
“At the end of that 15-year period, think about what will the landscape will look like when the product is on the decline? That gives you some clue as to where things might be going.”
On the capacity side, the planning lead times are longer than most companies assume. A workforce can flex plus or minus 10% within current capacity. Bringing in new staff takes around nine months to get operational, and closer to a year to reach full effectiveness. Capacity expansion runs about two years, and a completely new plant is a four-year commitment.
“You really want to be thinking at the four-year level, rather than the immediate level.”
On scenario planning, Nick offers a clear-eyed example using the Trump tariffs as a case study.
“Think through two very different scenarios. One: the tariffs are enacted, factories are rebuilt in the US, and there isn’t a brain drain. The sheer depth of American science is incredible. A year on, US economic growth has not been affected as much as most economists thought. The second scenario: China becomes a leading source of origination, a brain drain from the US does occur, regionalization of CDMOs leads to a loss of economies of scale, and the EU resurges.”
Neither scenario will play out in pure form, but building both allows organizations to act decisively as signals emerge.
He applies the same logic to pharma pricing, long assumed to be immune to political pressure.
“If you look at the price of pharma in the US compared to anywhere else, it’s not just that it’s three to four times more. Almost no other industry has that same price gap. If you just step back, the price issue in the US was so glaringly strange from an economic point of view. You should be thinking through the consequences of what happens if politicians get serious about it.”
Scenario planning, Nick concludes, isn’t about predicting the future. It’s about being ready for more than one version of it.
Nick Wigdahl be at CDMO Live Europe in Rotterdam, May 19 to 21, 2026. To get in touch, visit pharmgro.co.uk or email ni**@*********co.uk.










