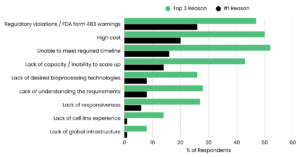
FDA Warning Surge: Regulatory Violations #1 Reason for CDMO Disqualification
Last week, we reported on the FDA’s accelerating enforcement activity — including 30 warning letters issued to telehealth companies in a single day and a broader surge in manufacturing-related citations. The response from the PharmaSource community was significant. A recurring









