Guest Editorial by Molly McGaughan, Co-founder and Managing Director, Firm
Contract development and manufacturing organizations sit much closer to the patient experience than most people realize. Their decisions shape whether a dose arrives on time, whether rescheduling is possible, and how issues are communicated when something goes wrong. In radiopharma, where treatment windows are measured in hours rather than days, these operational touchpoints directly impact patient access to potentially life-saving therapies.
Yet most CDMOs still operate with mindsets and systems built for traditional pharmaceutical manufacturing. This disconnect creates what we call “the software cliff”, and it represents the single biggest risk during commercial scale-up.
The Experience Economy Reaches Pharma Manufacturing
Pharmaceutical companies are increasingly differentiating through experience: speed, reliability, transparency, and digital engagement. This shift affects what they need from CDMO partners.
CDMOs are no longer just capacity providers. They are experience enablers. When pharma companies design services that promise predictability, responsiveness, and clarity to sites and clinicians, that promise can only be kept if CDMO partners operate with the same mindset. This means aligning on technical quality, service-level behaviors, data sharing, software standards, and responsiveness that support those experience goals.
The Software Cliff: Where Traditional Models Break Down
The biggest friction comes from trying to apply a standard “made-to-order” model to complex therapeutic isotopes like Lutetium-177 or Actinium-225. In traditional pharma, you receive an order, and then you manufacture. In radioligand therapy, the supply chain is so tight that production often needs to start before the patient’s order is even received.
This creates a massive gap in visibility and coordination. Pharma teams need a CDMO that can manage this paradox: orchestrating a manufacturing run based on forecasts, while simultaneously holding guaranteed “capacity slots” that can be instantly matched to a specific patient the moment an order arrives. Often, legacy CDMO systems cannot handle this decoupling of production from ordering. Manual handoffs that work effectively for traditional timelines can become strained under the pressure of a 2-day delivery window.
Compressed timelines remove any buffer for misalignment. A missed handoff, delayed data update, or unclear responsibility can directly translate into missed treatment windows. This reality forces a different level of integration between pharma and CDMOs. Shared operating models, shared data, and agreed escalation paths become fundamental to patient access.
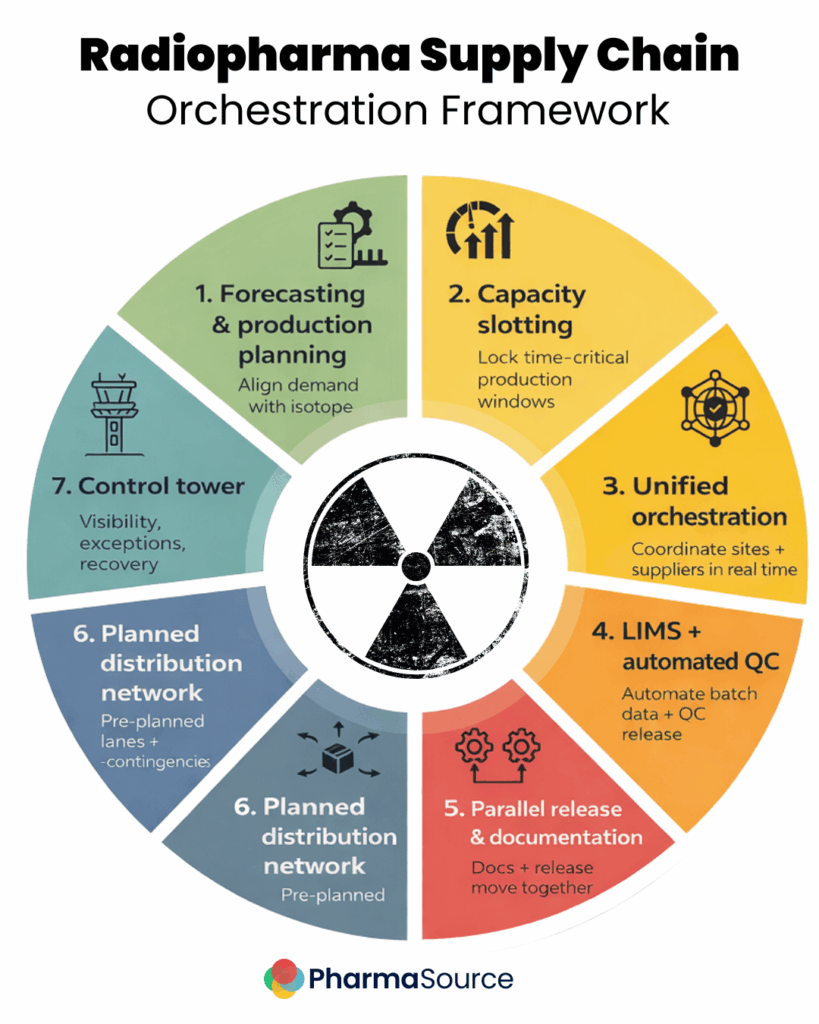
What a Well-Integrated CDMO Actually Looks Like
A well-integrated CDMO has moved beyond simple manufacturing to become a supply chain orchestrator. It requires digital infrastructure covering seven critical steps:
- Forecast & Production Planning: Production is decoupled from immediate orders. Systems must allow manufacturing to begin based on rolling forecasts and pre-booked capacity.
- Capacity Slotting: A “slot engine” guarantees capacity. When a center treats a patient, they claim a pre-validated manufacturing slot.
- Unified Orchestration: Whether the CDMO provides the portal or connects to yours, everything (production, QC, and logistics) must be driven by a single source of truth.
- LIMS & Automated QC: Quality Control cannot be a bottleneck. Integration must allow for automatic result processing and rules-based batch release.
- Parallel Release & Documentation: The product must be releasable while the shipment is being prepared. Automatic QC release triggers “ready to ship” status while the dose is effectively still in quarantine, saving critical hours.
- Planned Distribution Network: Transport cannot be an estimate. It requires pre-defined, time-validated routes with SLAs for every specific site and region.
- The Control Tower: A real-time view integrating ordering, manufacturing, and logistics. If a batch fails or a flight is delayed, the system must visualize it instantly so teams can re-allocate or re-route while there is still time to save the dose.
Two Paths to Interoperability
CDMOs need to stop thinking of their software as a closed loop and start thinking of it as an interoperable service. This manifests in two distinct ways:
- The “Full Stack” Partner: For biotechs or companies without their own logistics infrastructure, the CDMO provides the full ordering platform – a white-labeled portal where sites order, track, and manage patients directly on the CDMO’s infrastructure.
- The “Headless” Partner: For large pharma companies with their own Salesforce or SAP orchestration layers, the CDMO must act as a “headless” factory. MACH principles (Microservices, API-first, Cloud-native, Headless) are non-negotiable. The CDMO must expose its capacity, slotting, and status updates via API, allowing the pharma company to plug the CDMO directly into their own central operations without brittle workarounds.
Whether providing the front-end or just the back-end connectivity, the principle is the same: the CDMO’s internal operations must be transparent, digital, and adaptable to the user’s ecosystem.
The Competitive Advantage of Modernization
For CDMOs that continue to rely on fragmented, legacy systems, the risk is becoming a bottleneck. Fragmented systems slow response times, obscure root causes when issues arise, and make it harder to collaborate transparently with pharma partners. Over time, this erodes trust and limits the longevity of the partnership.
Conversely, modernization enables CDMOs to move upstream in value. They can support faster launches, smoother scale-up, and more resilient operations. For pharma partners, this translates into confidence and differentiation in the market. For CDMOs, it creates stickier relationships, earlier involvement in programs, and a reputation as a strategic partner rather than a transactional supplier.
The Essential Mindset Shift
Looking ahead, CDMOs will become more integrated ecosystem partners, playing a role not just in manufacturing but in shaping launch readiness, service models, and operational strategy.
The critical shift is from near-term outputs to long-term outcomes. Success isn’t just whether a batch was produced, but whether the system reliably enabled a patient to receive therapy as intended. CDMOs that design operations around that patient outcome will remain valued, indispensable partners in the radiopharma ecosystem.
Molly McGaughan is Co-founder and Managing Director at Firm, a consultancy dedicated to accelerating the delivery of advanced therapies. With 15 years of experience leading research, strategy, design, and technology programs, Molly partners with pharmaceutical companies to design customer-facing digital services and build the internal capabilities required to deliver advanced therapies at scale.