Your guide to choose the right Cell and Gene Therapy CDMOs for your company
The emerging world of cell and gene therapy (C>) is rapidly changing healthcare by using living cells and genetic modifications to treat diseases that were difficult to treat before.
At a time when 90% of biotechs rely on contract development and manufacturers, CDMOs play a crucial role in early stage and commercial launch of your products.
The cell and gene therapy CDMO market has become a critical pillar of the biopharmaceutical industry, fueled by rising demand for personalized, regenerative, and precision medicine. CDMOs provide the specialized process development, scale-up, analytical testing, GMP manufacturing, and regulatory support needed as CGTs move from clinical development to commercial production.
Because CGTs require sensitive handling, strict cold-chain logistics, and contamination-free environments, CDMOs offer the advanced infrastructure many biotechs and pharma companies lack in-house. Momentum continues to build, supported by increasing clinical trials, supportive regulatory pathways, and landmark approvals such as Luxturna and Zolgensma.
With growing investment in CGT R&D and more therapies progressing toward commercialization, the global CDMO landscape is rapidly evolving—marked by technological innovation, strategic partnerships, and rising demand for scalable manufacturing capacity.
In the latest PharmaSource podcast episode, Joshi Venugopal, Head of Region Europe for Novartis Gene Therapy & Rare Diseases shares valuable insights on how to successfully commercialise gene therapies in Europe, including innovative pricing models and critical success factors. His strategic approach offers essential guidance for biotechs and pharmaceutical companies looking to navigate the complex European landscape.

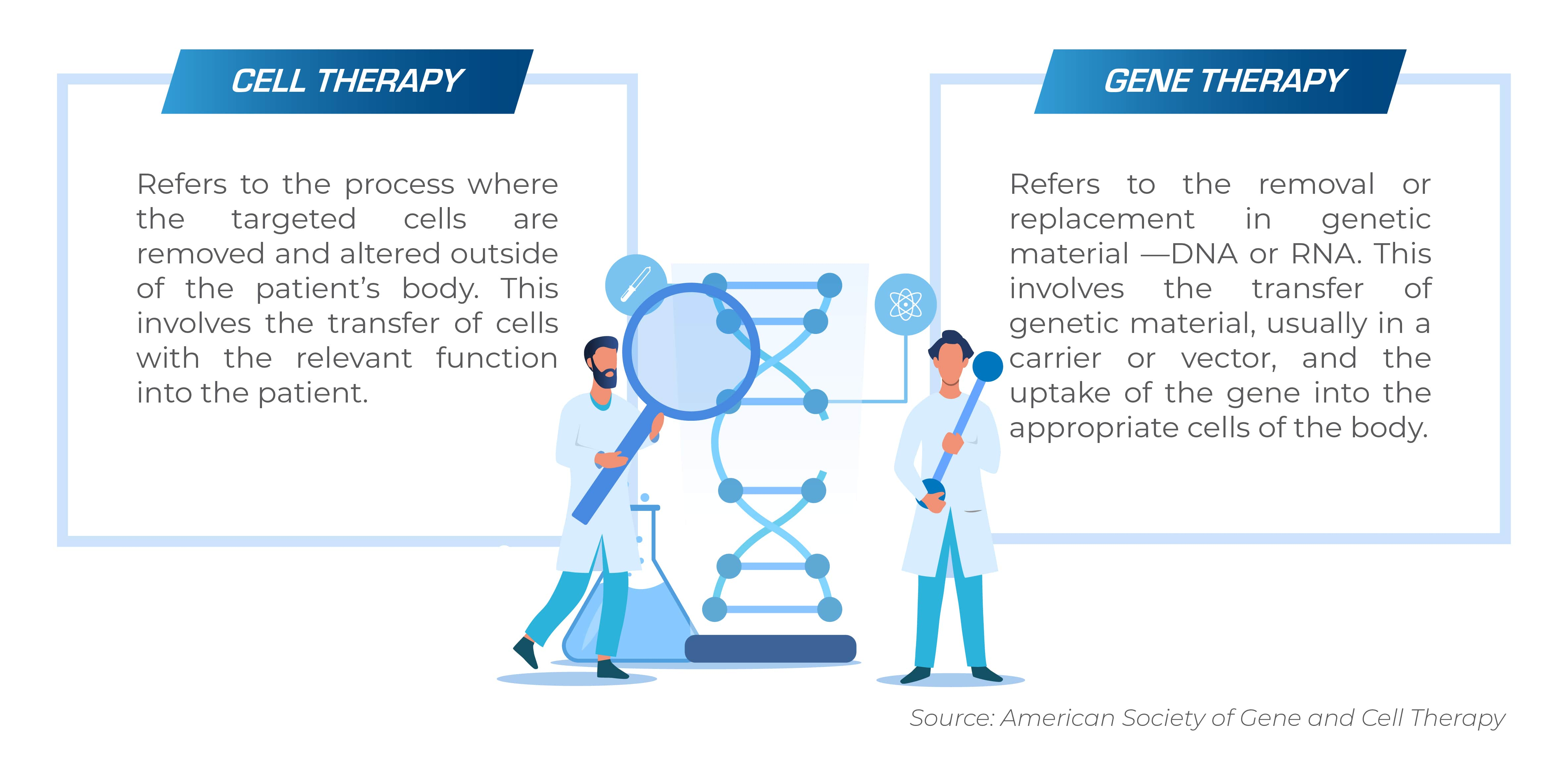
Understanding Cell and Gene Therapy
Cell therapy and gene therapy are overlapping fields of biomedical research and treatment. Both therapies aim to treat, prevent, or potentially cure diseases, and both approaches have the potential to alleviate the underlying cause of genetic diseases and acquired diseases. But, cell and gene therapies work differently.
Cell therapy aims to treat diseases by restoring or altering certain sets of cells or by using cells to carry a therapy through the body.
Gene therapy aims to treat diseases by replacing, inactivating or introducing genes into cells— either inside the body or outside of the body.
Some therapies are considered both cell and gene therapies. These therapies work by altering genes in specific types of cells and inserting them into the body.
Despite the long-standing similarities in traditional drug categories like small molecules, the US FDA and the EU EMA have yet to align their guidance on emerging therapies. This includes Cell and Gene Therapies (CGTs) and their EU equivalent, Advanced Therapy Medicinal Products (ATMPs).
Download the full e-book to understand the key differences between the US and EU in the Cell & Gene Therapy development and review processes.
Cell and gene therapy CDMOs
Cell & Gene Therapy CDMOs work as specialised extension of a biopharma’s team, giving you their knowledge, facilities, and tools to handle important parts of your C> development and manufacturing.
They cover everything from figuring out the process and making DNA to producing viral vectors and processing cells. They provide a complete set of services to make your journey from the early stages to delivering treatments as smooth as possible.
Subcategories of C> CDMOs:
The diverse C> CDMO landscape can be segmented based on service scope and expertise:
- Full-service C> CDMOs: These one-stop shops offer a complete suite of services encompassing preclinical to commercial-scale manufacturing.
- Niche C> CDMOs: These specialized players focus on specific areas like viral vector production, cell line engineering, or fill-finish operations.
- Emerging C> CDMOs: These new entrants offer innovative technologies and disruptive business models, often focusing on specific cell or gene therapy modalities.
Cell and Gene Therapy CDMOs Market Trends
The market is rapidly expanding with new cell and gene therapy products available, as researchers are focusing more on rare diseases.
The global cell and gene therapy CDMO market was valued at USD 4.31 billion in 2024 and is expected to reach USD 27.12 billion by 2033, reflecting a strong CAGR of 23.03% from 2025 to 2033. Growth is being fueled by an expanding pipeline of cell and gene therapy products, rising focus on rare diseases, and significant public and private investment in research and development. (Source – Grandview Research)
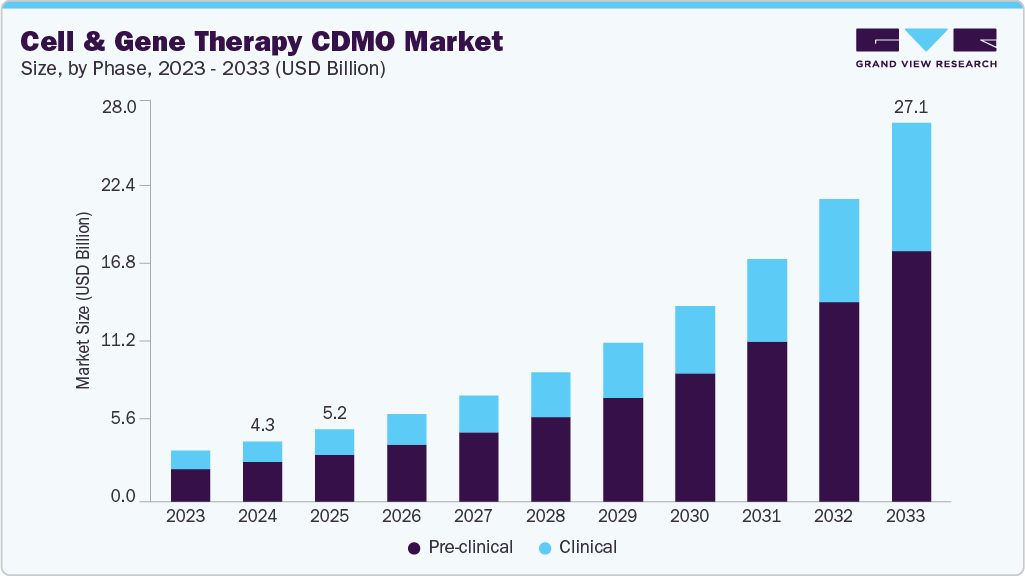
Source – Grandview Research
Key Growth Factors Driving the Cell and Gene Therapy CDMO Market
Innovative Technology:
CDMOs are rapidly investing in advanced cell and gene therapy technologies, including continuous manufacturing and AI-driven process optimization. These innovations enhance efficiency, scalability, and reliability—critical in the complex CGT development landscape.
Market Consolidation:
The sector is seeing increased consolidation as larger CDMOs acquire smaller firms to expand technical capabilities and geographic presence. This creates a more concentrated market with broader service offerings, greater operational scale, and improved efficiency for sponsors.
Geographical Expansion:
Global demand for CGTs is prompting CDMOs to expand into high-growth regions by strengthening infrastructure, increasing R&D investments, and aligning with supportive regulatory ecosystems. This wider footprint enables faster, more flexible supply chains.
Evolving Regulatory Landscape:
With regulatory frameworks for cell and gene therapies rapidly advancing, CDMOs are working closely with agencies to meet stringent, evolving requirements. Strong regulatory alignment is becoming a key differentiator in ensuring compliant and commercially viable CGT manufacturing.
Cell & Gene Therapy CDMOs Infographic
The number of CDMOs offering cell & Gene Therapies is expanding rapidly. This infographic summarises those operating facilities in Europe.

Download the European CDMO infographic here and the North American CDMO infographic here
Market Dynamics
Drivers – Growth in Clinical Trials for Innovative Therapies
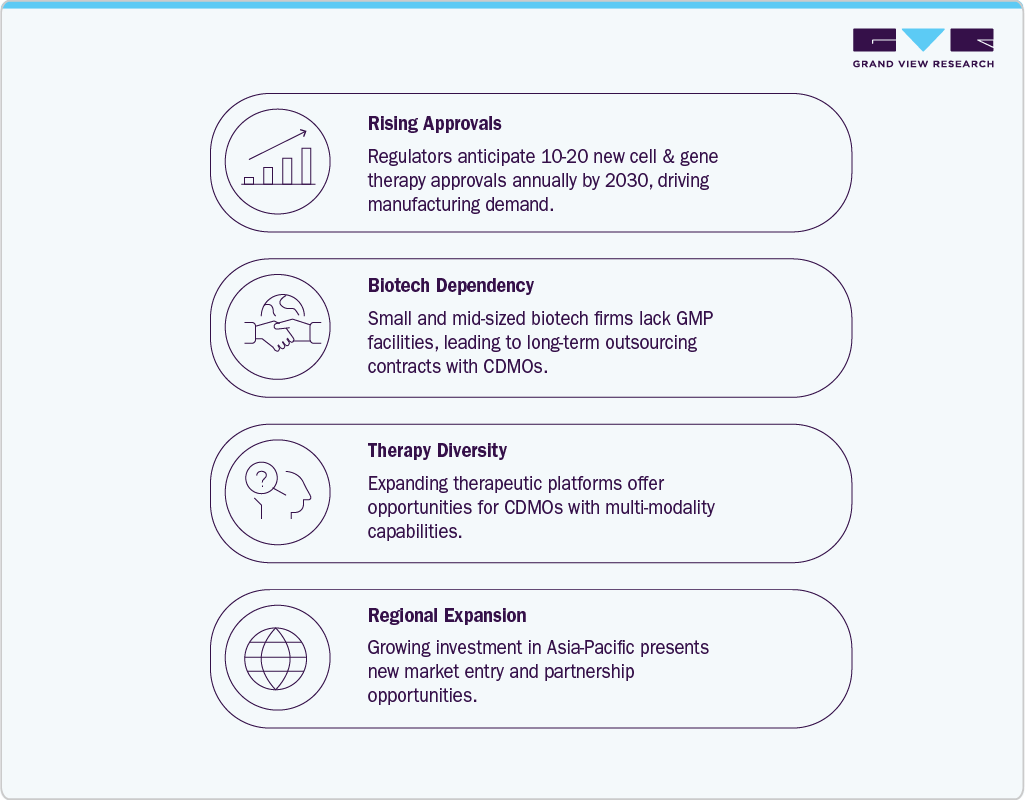
Cell and gene therapy (CGT) is expanding rapidly as living cells and genetic modification are increasingly used to treat complex diseases. CDMOs play a vital role in supporting this growth by providing clinical-grade materials, process development, and manufacturing expertise. With over 5,000 gene therapy trials listed by the NIH, demand for specialized CDMO support is rising. CDMOs not only supply materials for early-stage studies but also help scale successful therapies for commercial manufacturing, ensuring consistent quality through continuous process monitoring and optimization.
Restraint – High Manufacturing Complexity
The CGT CDMO market faces significant challenges due to the complexity of producing therapies that rely on live cells, viral vectors, and highly individualized treatment protocols. These products require rigorous quality systems, specialized infrastructure, and expert talent—factors that increase operational difficulty and drive up production costs. As a result, manufacturing complexity remains a key barrier to wider CGT adoption.
Opportunity – Access to Specialized Facilities and Advanced Technologies
Strong investment is flowing into specialized CGT facilities and cutting-edge technologies, positioning CDMOs as essential partners for development and manufacturing. Over the next five years, the sector will benefit from advancements in scalable production platforms, strategic collaborations, and accelerated drug discovery. While capacity constraints may persist, demand is expected to grow for CDMO capabilities supporting therapies for more common conditions, such as Parkinson’s disease, prompting expansion of large-scale development and manufacturing infrastructure.
Benefits of Partnering with C> CDMOs:
Working with Cell and Gene Therapy CDMOs can bring several advantages:
- Expertise and experience: Gain access to specialized knowledge and proven track records in C> development and manufacturing.
- Infrastructure and resources: Leverage state-of-the-art facilities, technologies, and equipment unavailable internally.
- Efficiency and speed: Accelerate your development timelines and optimise manufacturing processes through CDMO expertise.
- Cost optimisation: Access economies of scale and potentially reduce development costs compared to building internal capabilities.
- Regulatory compliance: Gain support from CDMOs experienced in navigating the evolving C> regulatory landscape.
Challenges of Partnering with C> CDMOs:
Cell and Gene Therapy CDMOs can come with some challenges:
- Selecting the right partner: Careful due diligence and evaluation are crucial to finding a CDMO that aligns with your specific needs and expertise.
- Intellectual property considerations: Clear agreements and confidentiality measures are essential to protect your intellectual property.
- Quality control and data integrity: Maintaining quality standards and data integrity requires robust oversight and communication with your CDMO partner. Integration and communication: Seamless integration of the CDMO into your development process and consistent communication are vital for project success.
- Integration and communication: Seamless integration of the CDMO into your development process and consistent communication are vital for project success.
Take a look at PharmaSource’s expert insight, which explores how cell and gene therapies (CGTs) are transforming treatment for genetic diseases, cancer, and other serious conditions—while introducing complex manufacturing challenges. Industry leaders share practical guidance on overcoming key obstacles such as scalability, quality control, and evolving regulatory requirements, offering a clear roadmap for advancing CGT development and production.

Opportunities
Collaborating with Cell and Gene Therapy CDMOs offers various opportunities for innovators:
- Expertise and experience: Cell and gene therapy CDMOs have a deep understanding of the complex processes involved in developing and manufacturing these therapies. They can provide your company with the expertise and experience you need to overcome challenges and bring your therapy to market faster.
- Access to specialized facilities and technologies: Cell and gene therapy CDMOs invest heavily in specialized facilities and technologies that are essential for developing and manufacturing these therapies. Partnering with a CDMO can give you access to these resources without having to make a significant upfront investment yourself.
- Improved efficiency and scalability: Cell and gene therapy CDMOs have optimised their processes for efficiency and scalability. This can help you to reduce your costs and bring your therapy to market faster.
- Reduced regulatory burden: The regulatory landscape for cell and gene therapies is complex and ever-changing. Cell and gene therapy CDMOs have the expertise to help you navigate this landscape and ensure that your therapy meets all regulatory requirements.
- Focus on your core competencies: By partnering with a cell and gene therapy CDMO, you can free up your internal resources to focus on your core competencies, such as drug discovery and development.
- Cost savings: In some cases, partnering with a cell and gene therapy CDMO can be more cost-effective than building your own manufacturing capabilities. This is especially true if you are a small or mid-sized company with limited resources.
- Faster time to market: By partnering with a cell and gene therapy CDMO, you can accelerate your development timelines and bring your therapy to market faster. This can be critical for therapies that are needed to treat patients with serious or life-threatening diseases.
Finding the Right C> CDMO Partner:
Selecting the right C> CDMO is a critical decision. Follow these steps for a successful partnership:
Define your needs and objectives: Clearly identify your specific project requirements, desired outcomes, and timeline.
Research and shortlist potential CDMOs: Conduct thorough research, considering their expertise, experience, facilities, technologies, and track record.
Develop a Request for Proposal (RFP): Outline your project details, requirements, evaluation criteria, and quality standards in a detailed RFP.
Evaluate proposals carefully: Assess each proposal against your evaluation criteria, focusing on relevant expertise, experience, cost structure, quality systems, and compliance record.
Conduct due diligence: Verify the CDMO’s financial stability, regulatory compliance, and infrastructure.
Negotiate and finalise: Approach negotiation with a collaborative mindset, seeking mutually beneficial solutions that align with both parties’ goals.
By carefully considering these factors, you can identify a C> CDMO partner that aligns with your project goals and contributes to the success of your cell and gene therapy development.
Latest C> CDMO News-
Kincell Bio Partners with Duke University to Advance Cell and Gene Therapy (Oct 2025)
Lonza Launches TheraPEAK® Products to Boost Cell and Gene Therapy Manufacturing (Oct 2025)
Made Scientific and Streamline Bio Validate AI-Driven Robotics for Cell Therapy Manufacturing (Oct 2025)
eXmoor Pharma and Siam Bioscience to Launch CGT Centre in Bangkok (Sep 2025)
Cellular Origins Secures £1M Innovate UK Grant for Automated CGT Manufacturing Platform (July 2025)
Cellular Origins and Thermo Fisher Launch Robotic CGT Manufacturing Platform (June 2025)
Cellex Cell Professionals Marks Decade in CGT with Major GMP Facility Expansion (June 2025)
Matica Biotechnology Partners with NCTM to Launch End-to-End CGT Manufacturing Platform (May 2025)
AGC Biologics Launches Cell and Gene Technologies Division for Advanced Therapies (April 2025)
The Federal Government of Canada Backs OmniaBio’s Expansion to Strengthen Cell and Gene Therapy Manufacturing (March 2025)
ENCell Signs MOU with Cell Resources to Expand CGT CDMO Operations in Japan (March 2025)
Rentschler Biopharma Ends Cell and Gene Therapy Work, Strengthens Biologics Focus (Jan 2025)
Download our CDMO News Tracker to stay ahead of every shift in the CDMO landscape.
















