An overview of the Contract Research Organisation (CRO) market and its significance in the pharmaceutical and biotech industries.
This guide provides you with the overall market size and trends, key drivers, opportunities and challenges, key suppliers in the category and how to improve strategic supplier partnerships.
The pharmaceutical and biotech industries are witnessing rapid growth and innovation, driving the need for efficient research and development (R&D) processes. Contract Research Organisations (CROs) have emerged as key partners providing outsourced services across various domains such as preclinical services, clinical trials management, regulatory affairs, and more.
Contract Research Organisations, are external service providers that offer specialised support to pharmaceutical and biotech companies. They play a critical role in accelerating drug development, reducing costs, and enhancing operational efficiency for their clients.
Breakdown of main services offered by CROs:
1. Preclinical Services
Preclinical testing, including in vitro and in vivo studies, to assess the safety and efficacy of drug candidates before human trials.
2. Clinical Trials Management
Planning, execution, and oversight of clinical trials, ensuring adherence to protocols, data collection, and regulatory compliance.
3. Clinical Data Management
Collection, validation, and analysis of clinical trial data, ensuring accuracy and compliance with regulatory standards.
4. Biostatistics and Statistical Analysis
Application of statistical methods to analyze clinical trial data, providing insights on efficacy, safety, and patient outcomes.
5. Regulatory Affairs
Expertise in navigating complex regulatory requirements, ensuring compliance with local and international regulations.
6. Pharmacovigilance and Drug Safety
Monitoring, evaluation, and reporting of adverse events and side effects associated with investigational drugs and marketed products.
7. Medical Writing
Preparation of regulatory documents, study protocols, investigator brochures, and other scientific and medical documents.
Contract Research Organisation Market Trends
The global contract research organization (CROs) services market size is calculated at USD 77.00 billion in 2025 and is predicted to increase from USD 82.90 billion in 2026 to approximately USD 149.34 billion by 2034, growing at a CAGR of 7.61% from 2025 to 2034.
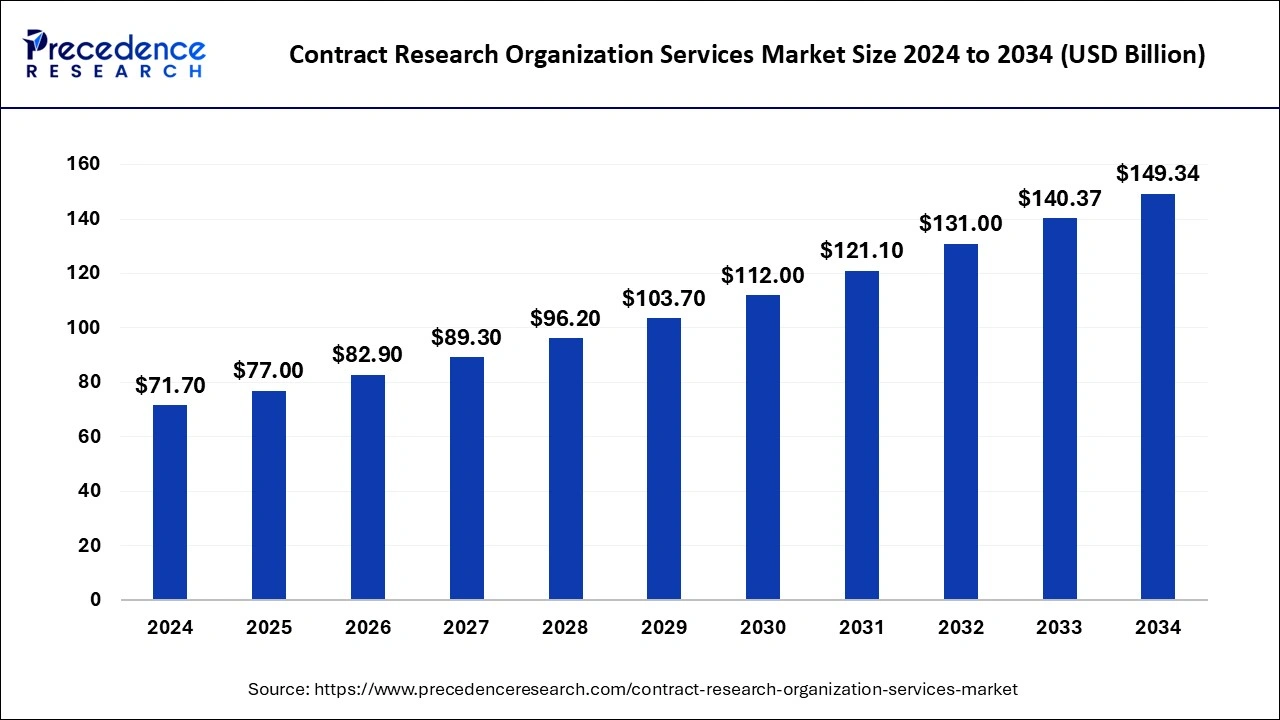
Source- Precedence Research
Market trends and key drivers
1. Increasing Outsourcing of R&D Activities
Pharma and biotech companies are outsourcing a larger portion of their R&D activities to CROs to leverage their expertise, reduce costs, and accelerate drug development timelines.
2. Growing Focus on Specialised Therapeutic Areas
There is a rising demand for CROs with expertise in specialised therapeutic areas, such as oncology, neurology, rare diseases, and gene therapies.
3. Technological Advancements and Data-Driven Approaches
CROs are adopting advanced technologies, such as artificial intelligence (AI), machine learning (ML), and big data analytics, to enhance efficiency, data quality, and decision-making capabilities.
4. Regulatory Complexities and Compliance Requirements
The pharmaceutical industry is subject to stringent regulatory frameworks, necessitating CROs to maintain expertise in regulatory affairs and ensure compliance throughout the drug development process.
5. Global Expansion and Emerging Markets
CROs are expanding their presence in emerging markets, such as Asia-Pacific and Latin America, to tap into growing opportunities and leverage cost advantages.
SWOT Analysis-
Strengths
Expertise in precision medicine, genomics, and biomarker-driven research
Strong adoption of digital tools, wearables, and remote trial technologies
Operational flexibility to support diverse and complex clinical programs
Weaknesses
Heavy dependence on sponsor pipelines and project-based revenues
High competition requiring continuous differentiation
Revenue instability without consistent new project inflow
Opportunities
Expansion of global clinical trials and broader patient access
Entry into emerging markets
Growing demand for CRO support in advanced therapies (gene therapy, immuno-oncology)
Threats
Sponsor budget cuts or pipeline shifts
Increasing competition from global CROs
Complex multi-regional regulatory requirements
Rapid technological evolution challenging slower adopters
How to partner better with Contract Research Organisations
In order to establish better partnerships with PAT suppliers and effectively manage costs without compromising quality, pharmaceutical procurement teams can consider the following strategies:
Clearly Define Project Requirements and Expectations
Clearly communicate project goals, timelines, deliverables, and quality expectations to ensure alignment between the pharmaceutical company and the CRO.
Foster Open Communication Channels
Establish regular communication channels, such as project meetings and status updates, to foster collaboration, address concerns, and ensure transparency.
Establish Performance Metrics and Key Performance Indicators (KPIs):
Define measurable performance metrics and KPIs to monitor the CRO’s performance, track progress, and address any deviations from agreed-upon standards.
Conduct Thorough Cost-Benefit Analyses
Evaluate the value proposition of engaging with a CRO by conducting comprehensive cost-benefit analyses, considering factors such as expertise, quality, timelines, and cost.
Explore Flexible Pricing Models and Contracts
Negotiate flexible pricing models, such as milestone-based payments or risk-sharing arrangements, to align incentives and optimize cost structures.
Streamline Processes and Eliminate Inefficiencies
Collaborate with CROs to identify and streamline inefficient processes, eliminating redundancies, and optimizing resource utilization.
Leverage Technology and Automation
Embrace technological advancements and automation solutions to improve operational efficiency, data quality, and cost-effectiveness.
Develop Long-Term Partnerships for Cost Optimization
Establish long-term partnerships with preferred CROs to leverage economies of scale, improve cost predictability, and foster continuous process improvement initiatives.

Source- Crown Bioscience
Recent CRO News-
FAQs-
1. What is a Contract Research Organisation (CRO)?
A CRO is a specialised service provider that supports pharmaceutical and biotech companies with outsourced research and development activities, including clinical trials, regulatory support, data management, and laboratory services. They help sponsors accelerate development while reducing operational burden.
2. What key trends are driving the CRO market today?
Major trends include the rise of precision medicine, increased adoption of decentralised and digital clinical trial technologies, growing outsourcing by small and mid-sized biotechs, and the global expansion of clinical trials into new geographic regions.
3. What challenges do pharma companies face when working with CROs?
Common challenges include inconsistent performance across study phases, communication gaps, limited operational oversight, unexpected delays, and difficulty aligning internal expectations with CRO capabilities. Managing multiple CRO partners across regions can also add complexity.
4. How should procurement teams evaluate and select the right CRO partner?
Procurement should assess the CRO’s therapeutic expertise, past performance, global footprint, regulatory track record, technology capabilities, cost structure, and cultural fit. Reviewing case studies, KPIs, and operational processes is essential for selecting the best partner.
5. How is globalisation influencing CRO operations?
Globalisation enables CROs to run trials across diverse populations, access faster patient recruitment, and collaborate with a broader network of investigative sites. It also opens opportunities in emerging markets, though it requires strong regulatory and logistical coordination.
6. How can sponsors build stronger partnerships with CROs?
Strong partnerships are built on clear communication, well-defined roles, transparent expectations, shared risk, joint planning, and continuous performance monitoring. Long-term, strategic collaboration—rather than project-by-project engagement—often leads to better trial outcomes.















