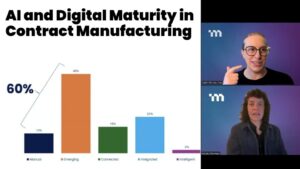
AI and Digital Maturity in Biopharma Contract Manufacturing: Key Findings from Research
Watch the presentation of the results with MasterControl

Watch the presentation of the results with MasterControl

In pharmaceutical manufacturing, the complexity of outsourcing relationships demands a systematic approach. As external manufacturing networks grow more critical to business success, organizations need a clear framework to guide their supplier management programmes. Each year, State of Flux’s Global SRM

As cell and gene therapies, RNA-based platforms, and antibody-drug conjugates accelerate toward commercialization, the pharmaceutical industry must develop analytical methods robust enough to characterize increasingly complex therapeutic modalities while remaining flexible enough to evolve with emerging science. PharmaSource asked analytical

As clinical development timelines compress and therapeutic complexity rises, pharmaceutical sponsors must secure manufacturing partnerships that adapt to uncertainty without draining budgets. How to structure CDMO contracts that deliver both flexibility and cost efficiency has become a strategic imperative and

In an era of persistent supply chain disruptions, how can pharmaceutical and biotech companies protect against single-source vulnerabilities without drowning in operational complexity? Six industry leaders recently weighed in on dual sourcing strategies, offering a roadmap for executives seeking resilient

MilliporeSigma is betting on advanced chromatography capabilities to address persistent bottlenecks in antibody manufacturing as the company prepares to integrate JSR Life Sciences’ Protein A resin technology into its bioprocessing portfolio. The acquisition, announced in October 2025 with an expected

At Nordic Life Science Days (NLSDays), leading pharmaceutical executives from Novo Nordisk, AstraZeneca, and Roche convened to discuss the evolving landscape of cardiovascular, renal, and metabolic (CVRM) drug development. The “Transforming CVRM Health” panel revealed that despite clinical successes with

The relationship between pharmaceutical companies and their manufacturing partners is changing. At CPHI Frankfurt 2025, industry leaders outlined a new approach to CDMO selection and management during the talk, “CDMOs at a Crossroads: Scaling, Adapting, and Partnering for the Future.”

Decarbonization is a business imperative requiring stronger collaboration among providers, manufacturers, and suppliers. The PSCI Decarbonization Summit 2025 brought together leaders across the pharma value chain to share strategies and to advance decarbonization. The closing panel featured Cristina Indiveri, AVP

Most pharma companies have climate commitments. Far fewer have woven those standards into how they select, onboard, and work with suppliers. At the PSCI Decarbonization Summit 2025, Koen Postuma, Director of Procurement at Merck, Vincent Barbotin, Scope 3 Active Ingredient
