This guest editorial was contributed by SmartSkin Technologies
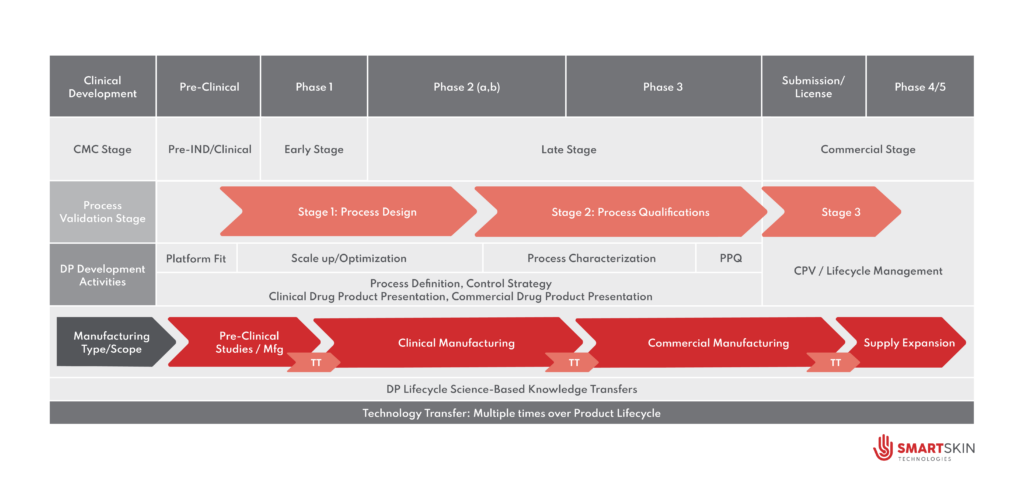
Technology transfer is a quintessential part of the CMC (Chemistry Manufacturing and Control) along the pre-commercial, commercial, and post-commercial phase of a pharmaceutical product life cycle. Critical to the success of the transition to the next phase of development and supply of materials in clinical or commercial phase, technology transfer takes place between a sending site and a receiving site selected by the product owner in alignment with their strategic and business intent.
During clinical development, when a sponsor entrusts a process to a CDMO for manufacturing of clinical materials aligned with the desired Target product profile (TPP), the sending team is not simply handing over the current knowledge related to the product and the processes associated with the way to manufacture and control the desired batches. They are handing over the responsibility for creating a process at scale with consistency, reliability, quality, and timely supply to the patients. For CDMOs, meeting those expectations requires more than executing a process accurately as known by the sending site. It requires demonstrating scientific understanding, operational excellence, discipline, and control to overcome the subtle gaps in knowledge, differences, and sources of variability between the sites, that influence the execution of every step in the process.
During technology transfer, both product-specific knowledge (i.e., molecule attributes, product dosage form, product specification, stability) and process-specific knowledge (i.e scale, site equipment, sterile processing operations integrated with product control strategy) must be shared between sites, understood, and appropriately evaluated for potential gaps. During the initiation of the technology transfer, the gaps should be identified and discussed between the sending and receiving teams. Transfer activities and their associated studies will aim to eliminate gaps based on their risks to the success of the upcoming milestones.
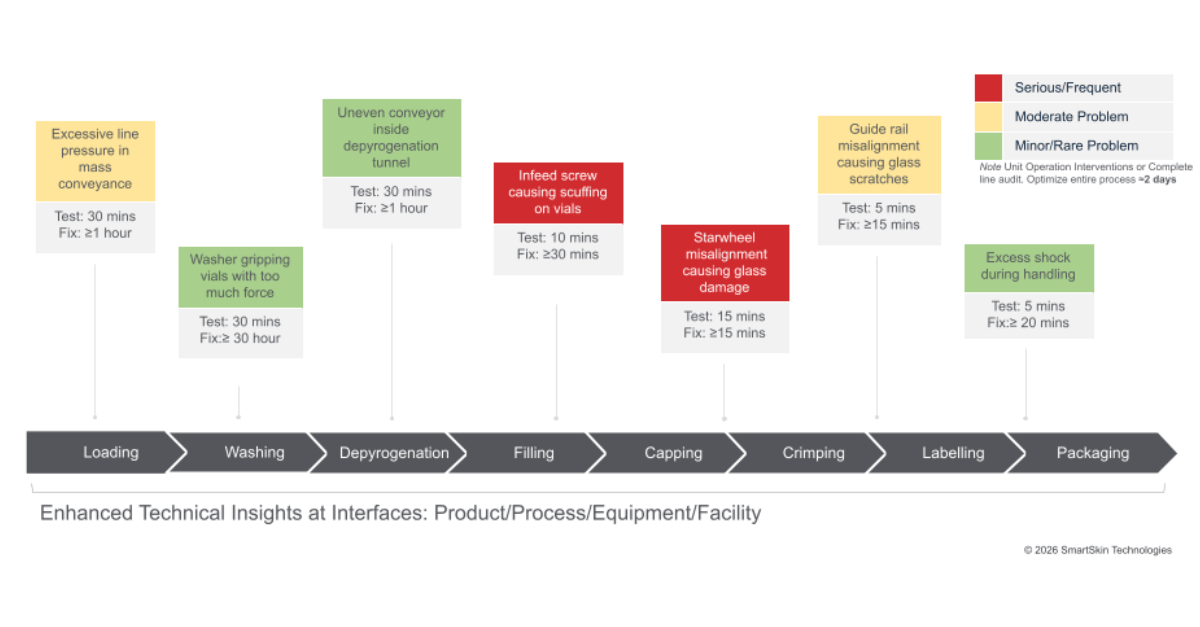
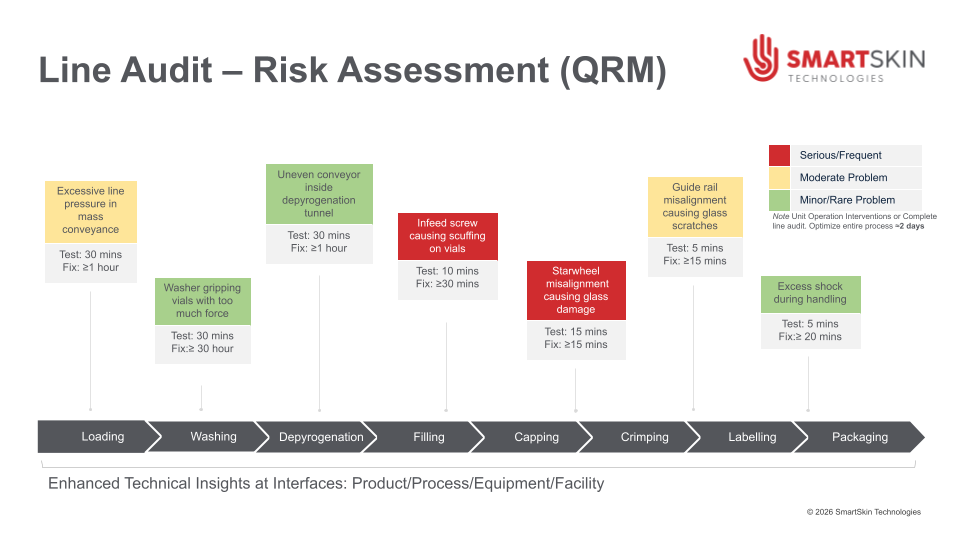
Across clinical and commercial manufacturing of injectable products, one of the persistent barriers to predictable technology transfer outcomes is the hidden gaps and variability created by the differences in equipment trains. The unknown or uncharacterized operating conditions generated at the interface between the product and equipment at the receiving site become a source of “surprises”, delays, repeated activities, and unexpected modifications impacting the TT (Technology Transfer) timing. Equipment design, mechanical, handling, and equipment-driven forces that shape how a container moves through a fill-finish, inspection, or packaging line often go unmeasured and understood. Yet they are deeply consequential for the outcomes expected from the receiving site.
Understanding them is becoming essential for CDMOs operating at the intersection of development, scale-up, and market readiness.
One example of how CDMOs are addressing this challenge is by adopting technologies that make the physical interactions between equipment and product visible and measurable in real time. Rather than relying solely on indirect indicators or post-event inspection, these approaches allow manufacturers to characterize how containers are actually handled as they move through filling and finishing operations.
Several technologies now exist to address this challenge, using instrumented surrogate containers that replicate the physical dimensions of the product presentation to measure and quantify the forces, impacts, and vibrations experienced at each handling interface in real time.
The Hidden Layer of Variability to Discover and Convert into Transferable Knowledge
Even if the drug product presentation and its assigned CQA remain unchanged during the transfer, the manufacturing environment experienced by the product filled into vials, syringes, cartridges, etc., always does. Differences in line design, equipment-specific technology, operating conditions, and the means to control them can alter how an injectable product is handled throughout production.
These small, often invisible differences influence:
- Container integrity
- Seal and closure performance
- Probability of defects, microcracks, or leaks
- Line stability and reject rates
- Downtime and yield
- Batch reliability at clinical and commercial scales
Machinability studies are the standard way to have a quick look at CDMO’s or receiving site equipment fit with a new product presentation. Visual observations and small adjustments can be made to initiate the introduction to the new site. However, this historically used practice does not measure the magnitude of interactions, their location, nor does it generate knowledge to be used towards establishing suitable operating parameters. Therefore, the transfer does not lead to a reliable, repeatable, predictable process; the risk of damage and interruption remains and manifests itself accordingly, especially as the scale of the production lots is increasing. To eliminate variation, avoid the events during production, and increase the product quality and decrease risks, CDMOs need to gather such processing knowledge that is measurable, digitalized, and available to characterize numerically at each step along the process, producing good outcomes.
Sponsors expect CDMOs not just to run the process; they expect them to understand it and defend it in front of regulators. That expectation intensifies as development progresses toward the late phase, where commercial readiness and regulatory scrutiny come into play.
Technology Transfer as a Scientific Exercise
A successful transfer requires the sponsor and CDMO to align on more than process instructions. They must align with the underlying scientific logic:
- Why were the parameters chosen
- How the product was handled during development
- Where the process showed sensitivity
- What Mechanical Conditions influence outcomes
- How line performance differed across phases
For CDMOs, treating technology transfer as a scientific endeavor creates value, trust, and confidence in documenting and deploying the scientific knowledge. It reduces assumptions, accelerates understanding, and establishes the foundation for consistent execution.
Making Mechanical Variability Visible
A growing number of CDMOs are focusing on understanding the physical forces that drive variation across lines, shifts, and facilities. When these forces are measured and analyzed, they reveal a clear picture of how the line interacts with the product.
Visibility enables teams to:
- Identify high-risk locations along the line
- Tune equipment precisely and consistently
- Reduce container damage and mechanical stress
- Understand why one site behaves differently from another
- Eliminate variability before engineering or commercial batches
- Provide evidence-based justification during regulatory review
This approach replaces trial-and-error adjustments with informed, data-driven decisions rooted in measured behavior rather than operator intuition.
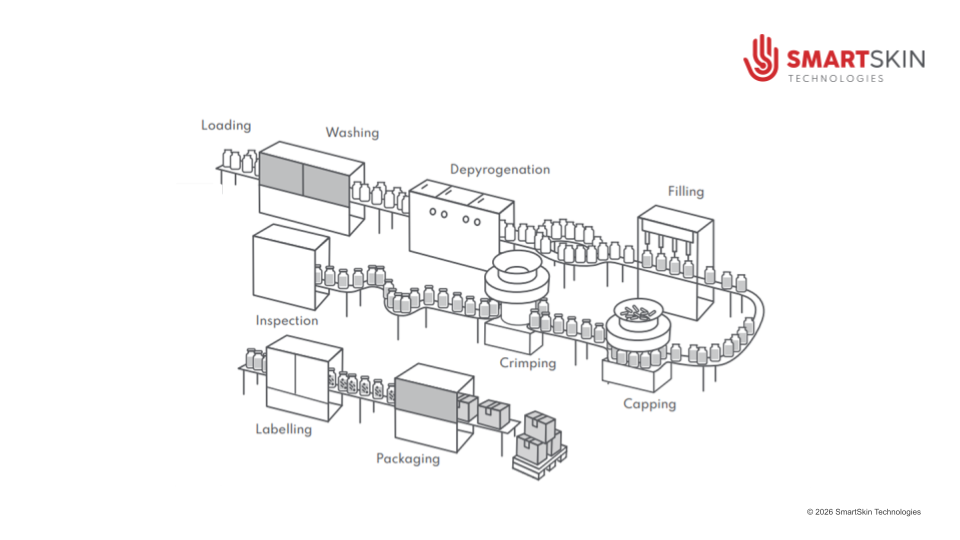
Pharmaceutical Fill-Finish and Packaging Process Overview
Establishing a Digital Baseline Before the Transfer
The digital era and the progress towards smart manufacturing are now enabling electronic transfers. More product and process data are electronically captured and become transferable, used for additional analytics and process capability assessments. One of the most powerful tools in modern technology transfer is the creation of a digital baseline, a quantitative reference profile of how a container should move through a well-configured line.
This baseline becomes a shared scientific language between sponsor and CDMO. It captures:
- The expected force ranges
- Acceptable mechanical variation
- Areas where the product is sensitive to equipment design
- The conditions under which the process was previously successful
With this baseline, CDMOs can evaluate their own equipment, identify differences immediately, and align their setup to the sponsor’s expectations before executing GMP batches. This not only reduces risk but also shortens the learning curve during scale-up.
For clinical manufacturing where material is limited and expensive, creating a baseline and reducing the visualized risks protects scarce supply. For commercial readiness, it builds reliability into the process before full volumes come into play, and confidence in the readiness for commercial transfer or PPQ. Such baselines and the associated process knowledge are expediting implementation of changes to the existing processes (equipment changes, adding or changing suppliers of packaging materials), and can be highly valuable to compare performance across lines used for the same product presentation
From Reactive Corrections to Preventative Control
In many facilities, mechanical issues historically surface only when something goes wrong: a breakage, a leak, a deviation, or a batch investigation. By the time an issue is visible, the cost of investment in making the commitment to launch production starts, dealing with deviations, time and materials loss, risks to compromise product integrity within the batch, and product loss is already incurred. The majority of such events are now preventable. A preventative mindset flips this dynamic.
When CDMOs quantify line behavior early in the technology transfer cycle, they can:
- Detect subtle equipment issues before they affect product
- Eliminate sources of stress that lead to damage, major or critical defect microcracks or stabilize and characterize more extensively the processing conditions before start of a new batch
- Expedite line turnover, returning faster equipment to production after maintenance activities, initiation of validation activities etc.
- Reduce the frequency of deviations and enable faster, evidence-based root cause identification during investigations
- Increase the predictability of both clinical and commercial supply
This shift toward prevention reflects the direction in which modern manufacturing and regulatory expectations are evolving. Current regulations emphasize the need for science-based process knowledge, risk management, understanding, improved and integrated controls strategy across the product lifecycle control. Technology transfers and the way they are executed are paramount to the success of the business, but also in responding to regulatory expectations for science-based process understanding and control.
Strengthening Sponsor Relationships Through Scientific Knowledge and Transparency
Sponsors evaluate CDMOs not only on technical capability, but on how well they communicate. Transparency grounded in scientific data is one of the strongest ways to build confidence during technology transfer.
When CDMOs share with the sponsor tangible insights as seen in equipment/product interaction force profiles, baseline comparisons, line behavior, and risk assessments summary, they provide sponsors with clear evidence that the process is understood, controlled, and ready to scale.
This transparency transforms the relationship:
- From transactional to collaborative
- From interpretation to evidence
- From reactive problem-solving to shared foresight
It also creates a feedback loop where both parties can jointly refine the process, improving stability, cycle times, and product quality across the lifecycle.
Supporting Evolving Regulatory Expectations
Regulators are increasingly focused on ensuring manufacturers and CDMOs engaged in GMP activities demonstrate scientific understanding, not just procedural compliance. Lifecycle documentation, justification of process choices, clear knowledge transfer, and evidence of equipment readiness are central themes in recent guidance.
CDMOs that proactively characterize line behavior and variability are better positioned to support:
- Regulatory filings: INDs, BLAs, NDAs, Change management documents
- Qualification and validation documentation content
- Site inspections with consistent and science-based operations and compliance practices
- Comparability studies and change management activities
- Commercial scale-up on the same fill/finish lines or alternate commercial lines
A structured, data-driven approach to technology transfer not only strengthens science, but it also reduces risk during regulatory engagement.
Scientific Understanding as a Strategic Advantage
As the biopharmaceutical landscape grows more complex, CDMOs face rising expectations around quality, speed, and reliability. Those expectations cannot be met by documentation alone.
The CDMOs that will lead in the next decade are those that:
- Expose mechanical and handling variability
- Build rigorous digital baselines
- Understand process behavior across phases
- Embrace preventative control
- Communicate transparently with sponsors
- Treat knowledge as the most valuable asset in technology transfer
When CDMOs transfer knowledge along with products, they strengthen the scientific foundation of every program they support and build trust that endures well beyond the first batch.