Your guide to choosing the right partner for pharmaceutical quality control services
Medicine is the science and practice of maintaining health and preventing, curing disease. Before consuming any type of medicine, they need to be tested and approved for consumption in prepared pharmaceutical laboratories, so that they can be sold and consumed by the population. Keeping patients safe and following the rules set by the government are the most crucial things. Quality control services are like a guide in this complicated process. This guide is made especially for people who make decisions in pharmaceutical companies. It helps you know what to look for when choosing a supplier for your medicines. This way, you can protect your brand and make your process of creating and making drugs even better.
Pharmaceutical quality control is no longer just a compliance function — it is a strategic capability that determines whether drugs reach patients safely, on schedule, and at scale. For pharma and biotech companies navigating complex pipelines, outsourcing quality control to a qualified CDMO partner has become an essential part of modern drug development and commercial manufacturing strategy.
Understanding the pharmaceutical quality control
Quality control (QC) in the pharmaceutical industry is a set of procedures to ensure the identity and purity of a pharmaceutical product. It’s a fundamental aspect of the industry, ensuring the safety and efficacy of drugs and maintaining the integrity of the supply chain. The main objective of quality control in the Pharmaceutical Industry is the drug analysis and testing in their various stages of production, verifying that they are able to proceed to the next stage and release the manufacturing process in accordance with the FDA quality control regulations and specifications required for consumption.The pharmaceutical quality control services include many different parts, each playing an important role:
- Analytical testing: Ensures the purity, potency, and identity of materials and finished products using sophisticated analytical techniques.
- Microbiological testing: Safeguards against microbial contamination, protecting patients from potentially harmful pathogens.
- Environmental monitoring: Monitors air, water, and temperature conditions to guarantee a sterile and controlled production environment.
- Stability testing: Assesses the shelf life and degradation profile of drugs, ensuring efficacy throughout their lifespan.
- Packaging and labelling inspection: Verifies conformity to regulations and ensures accurate information reaches patients.
- Validation and calibration: Guarantees the accuracy and reliability of equipment used in testing and manufacturing processes.

Source: Tutor Aspire
Market trends and key drivers
Pharmaceutical quality control refers to the integration of all procedures undertaken to ensure the identity and purity of particular pharmaceutical products. The rise in awareness for quality control of pharmaceuticals products is helping the global pharmaceutical quality control market to grow at a rapid rate. The increase in the development of comprehensive analytical tests drives market growth. The demand for integrated analytical services spanning all stages of drug discovery, development, and manufacturing also drives the market growth to further height.
The global pharmaceutical quality control market size was valued at USD 2.81 billion in 2024 and is expected to reach USD 6.14 billion by 2032, at a CAGR of 10.23% during the forecast period.
Source: Data Bridge
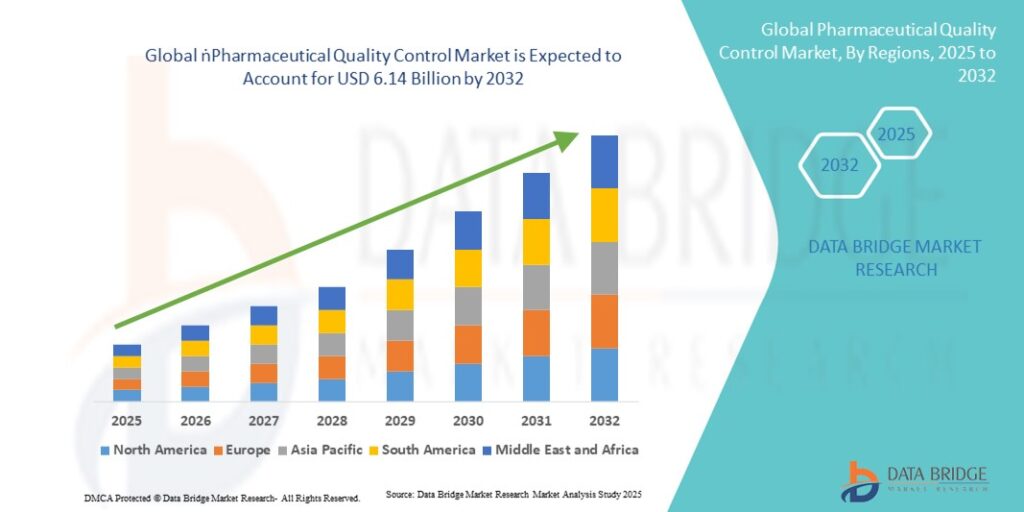
Three macro trends are currently reshaping pharmaceutical QC outsourcing-
First, the expansion of biologics and cell and gene therapy pipelines is increasing demand for highly specialized analytical capabilities that few sponsors can build internally.
Second, global regulatory convergence — particularly alignment between FDA, EMA, and PMDA expectations for data integrity and laboratory controls — is raising the baseline bar for what qualified QC outsourcing looks like.
Third, capacity constraints at commercial-scale manufacturers are pushing sponsors to seek CDMOs with integrated QC operations, rather than managing separate analytical and manufacturing relationships.
Key drivers
The market for quality control services is growing really fast because of some important reasons.
- Stringent Regulatory Requirements: The pharmaceutical industry is subject to strict regulations and quality standards imposed by regulatory bodies worldwide. Compliance with these regulations drives the demand for robust quality control measures
- Increasing Focus on Patient Safety: Patient safety is a critical concern in the pharmaceutical industry. Quality control ensures that drugs are safe, effective, and free from contaminants, ensuring patient well-being and trust in pharmaceutical products
- Growing Generic Drug Market: The expanding market for generic drugs necessitates stringent quality control to ensure bioequivalence and meet regulatory requirements. Quality control plays a crucial role in maintaining the safety and efficacy of generic drug formulations
- Sustainability in focus: Environmental regulations and consumer awareness are driving demand for eco-friendly quality control practices and waste reduction strategies.
Benefits
The advantages of partnering with good quality control service provider has lots of benefits.
Enhanced compliance
- Gain peace of mind with expert guidance on navigating complex regulations and maintaining impeccable quality standards.
Reduced risk of contamination and errors
- Minimise the risk of defective products reaching the market, protecting your brand reputation and patient safety.
Faster product development and launch
- Streamline processes and accelerate product approvals through streamlined testing and documentation.
Improved operational efficiency
- Leverage advanced technologies and expertise to optimise manufacturing processes and minimise waste.
Access to specialised expertise
- Gain access to cutting-edge analytical equipment and skilled personnel not readily available in-house.
Cost-effectiveness
- Outsource expertise instead of investing in expensive equipment and training, freeing up resources for core activities.
Challenges
- Complex Supply Chains: The pharmaceutical industry relies on global supply chains, making quality control challenging due to factors like transportation, storage, and distribution across various regions. Ensuring consistent quality throughout the supply chain is a significant challenge
- Analytical Method Validation: Validating analytical methods used in quality control is crucial for accurate and reliable results. Developing and validating robust methods for complex drug formulations and diverse matrices can be a challenge for pharmaceutical quality control laboratories
- Finding the right partner: Choosing the right quality control service provider is crucial. Factors like expertise, experience, technology capabilities, cost, and geographical reach need careful consideration to ensure a good fit for your specific needs.
- Cost and resource allocation: Setting up a thorough quality control system can cost a lot. It means spending money on fancy equipment, hiring skilled people, and keeping things running, which adds up over time. Figuring out how to manage these costs alongside other things we need in the company can be a bit tricky.
- Regulatory compliance: In the medicine business, the rules are always changing. We have to follow GMP compliance for pharmaceuticals like cGMP and ICH Q9. Keeping up with these rules is a bit tough for both our team inside the company and the ones we get help from outside.
- Managing communication and collaboration: Effective communication and collaboration between internal teams, external providers, and regulatory agencies are essential for successful quality control implementation. Establishing clear lines of communication and resolving conflicts can be challenging, especially in geographically dispersed or complex setups.
Opportunities
- Increasing Global Pharmaceutical Production: The growing demand for pharmaceutical products, driven by population growth and rising healthcare needs, presents opportunities for the expansion of quality control services to meet the rising production volumes
- Outsourcing of Quality Control Services: Many pharmaceutical companies outsource their quality control activities to specialized service providers. This trend creates opportunities for contract research organisations (CROs) and third-party laboratories offering quality control services
- Technological Advancements: The use of advanced technology in quality control presents opportunities to improve efficiency and accuracy in testing and analysis.
- Global Expansion: As pharmaceutical markets expand globally, there’s a need for quality control services to ensure consistency and compliance across different regions.
- Outsourcing: Companies looking to streamline operations may outsource their quality control needs, creating opportunities for service providers.
- Regulatory Changes: Changes in regulations often lead to new opportunities for services that help companies adapt and stay compliant.
- Data Management: With an increasing emphasis on data integrity, there are opportunities for services that specialise in secure and efficient data management.
- Customised Solutions: Tailoring quality control services to meet specific needs of pharmaceutical companies provides opportunities for niche service providers.
- Collaboration and Partnerships: Forming partnerships with pharmaceutical companies for comprehensive quality control solutions can be a strategic opportunity.
- Focus on Patient-Centric Quality: Services that prioritise patient safety and satisfaction align with the growing trend of patient-centric healthcare.
- Environmental Monitoring: As environmental considerations become more important, services focusing on monitoring and maintaining sustainable practices in pharmaceutical production have opportunities
This section features two insightful podcasts highlighting how leading pharma companies ensure quality across external manufacturing—Takeda’s approach to driving quality excellence and UCB’s leadership perspective shared by Liesbeth Foesters
CDMO Outsourcing for Quality Control Services: Selection Criteria and Best Practices
Outsourcing pharmaceutical QC testing to a CDMO requires careful evaluation of technical capabilities, regulatory track record, and operational fit. The right partner is not simply the one with the lowest per-test pricing — it is the one whose laboratory infrastructure, quality systems, and scientific expertise align with your product’s specific needs.
Technical and Operational Considerations
Method validation is a central consideration in any QC outsourcing relationship. The CDMO must be able to validate or transfer analytical methods to the standard required by the regulatory authorities in your target markets — whether that means ICH Q2(R2) compliance for analytical procedures or adherence to USP, EP, or JP compendial methods.
Turnaround time matters, particularly for time-sensitive batch release testing on commercial products or clinical trial materials. Understanding the CDMO’s standard turnaround commitments — and whether expedited options exist — is essential before committing to a partnership. Equally important is understanding their LIMS infrastructure: how data is generated, stored, reviewed, and protected against unauthorized alteration.
Key Criteria for Selecting a QC CDMO Partner
When evaluating a CDMO for quality control services, pharma and biotech professionals should assess the following:
- Regulatory certifications and inspection history. The facility should hold current GMP certification from the relevant regulatory authorities (FDA, EMA, Health Canada, etc.) and have a clean or well-resolved inspection record. Request a summary of recent inspection outcomes.
- Analytical platform breadth. Confirm the CDMO offers the specific techniques your product requires — HPLC, LC-MS/MS, Karl Fischer, dissolution, particle size analysis, bioassay, microbiology, etc. — and that instrumentation is current and well-maintained.
- Microbiology and sterility testing capabilities. For sterile and parenteral products, evaluate dedicated microbiology laboratories, environmental monitoring programs, and the scope of compendial microbiological testing available.
- Method development and validation expertise. Assess whether the CDMO can develop new methods or only execute existing ones, and review their validation track record across similar product types.
- Data integrity systems. Evaluate whether the CDMO’s LIMS and electronic laboratory notebooks (ELNs) meet 21 CFR Part 11 or EU Annex 11 requirements and whether audit trails are routinely reviewed.
- Stability program infrastructure. If long-term or accelerated stability studies are required, confirm the CDMO operates ICH-compliant stability chambers with continuous monitoring and alarm systems.
- Capacity and scalability. Understand current laboratory utilization rates and how the CDMO manages demand peaks. Ask whether dedicated capacity arrangements are available for high-volume or time-critical programs.
- Scientific and regulatory expertise. Evaluate the qualifications of QC leadership and regulatory affairs personnel, particularly their experience supporting product submissions in your target markets.
Red Flags to Watch for When Evaluating QC CDMO Partners
Several warning signs warrant heightened scrutiny during due diligence. A CDMO with a history of FDA Warning Letters or EMA GMP non-compliance findings related to laboratory controls is a serious concern. Similarly, high staff turnover in QC leadership roles can signal systemic cultural or operational problems that will affect your program.
Be cautious of partners who cannot provide specific examples of successful method transfers or who are reluctant to share inspection outcomes. Vague responses about LIMS capabilities, data backup protocols, or out-of-specification investigation procedures often indicate immature quality systems. Finally, be alert to CDMOs quoting unrealistically low turnaround times or pricing — both may reflect overpromising that creates problems at execution.
5 Questions to Ask Shortlisted QC CDMOs Before Signing
- Can you walk me through your standard method transfer or validation process, including timelines and deliverables at each stage? This reveals the depth of their procedural rigor and experience.
- What was the outcome of your last regulatory inspection, and how did you address any observations? A confident, transparent response signals a mature quality culture. Evasiveness does not.
- How do you manage out-of-specification results — what is your investigation process, and what access will we have to raw data? Understanding the OOS workflow protects you if a batch fails during your program.
- What is your current laboratory capacity utilization, and can you guarantee turnaround times for batch release testing under our projected volumes? Capacity commitments should be written into the quality agreement, not implied verbally.
- What experience does your team have with products similar to ours in terms of modality, route of administration, and target regulatory markets? Relevant experience shortens timelines and reduces risk significantly.
Recent Quality Control CDMO News-
Download our CDMO News Tracker to stay ahead of every shift in the CDMO landscape.
FAQs-
1. Why is pharmaceutical quality control so important?
Pharmaceutical quality control ensures that drugs are safe, effective, and free from contaminants before reaching patients. It maintains product integrity, complies with regulatory standards, and safeguards public health.
2. What are the key services included in pharmaceutical quality control?
These services typically include analytical and microbiological testing, environmental monitoring, stability testing, packaging inspection, equipment validation, and calibration to ensure every stage of the production process meets quality standards.
3. How can I assess the credibility of a quality control service provider?
Look for certifications, regulatory compliance (like cGMP and ICH Q9), client references, track record, experience in the pharmaceutical sector, and their technological capabilities. On-site audits and trial projects can also help validate their reliability.
4. What challenges should I be aware of when outsourcing quality control?
Challenges include managing communication across teams, ensuring regulatory compliance, handling complex supply chains, validating testing methods, and balancing cost with performance. A strong partnership with clear roles helps mitigate these issues.
5. What are the benefits of outsourcing quality control services?
Outsourcing offers access to specialized expertise and advanced technology, reduces operational costs, increases efficiency, speeds up product launches, and ensures stronger regulatory compliance, allowing internal teams to focus on core business activities.
6. What trends are shaping the pharmaceutical quality control industry today?
Key trends include the adoption of AI and automation, focus on data integrity, patient-centric quality measures, risk-based quality strategies, sustainable practices, and increased collaboration between pharma companies and QC service providers.










