An overview of the Intermediates manufacturing category for pharmaceutical procurement teams. This guide provides you with the overall market size and trends, key drivers, opportunities and challenges, contract manufacturers in the category and advice on how to improve strategic supplier partnerships.
Definition
Intermediates refer to chemical compounds that are used as building blocks or precursor materials in the synthesis of active pharmaceutical ingredients (APIs) or finished pharmaceutical products.
These intermediates play a crucial role in the pharmaceutical industry by enabling the efficient and cost-effective production of drugs.
Pharmaceutical intermediates can include various organic and inorganic compounds, such as reagents, catalysts, chiral compounds, and other functional groups, which are utilised at different stages of the drug manufacturing process.
Pharmaceutical intermediates serve as a critical link between the discovery of new drug candidates and the commercial production of pharmaceutical products. They allow for the synthesis of complex molecules and provide the necessary chemical transformations to convert raw materials into active drugs.
Pharmaceutical intermediates contribute to enhancing drug efficacy, stability, and bioavailability, thereby ensuring the quality and effectiveness of medications. These intermediates also play a vital role in achieving regulatory compliance by meeting the stringent standards set by regulatory authorities.
Market Size and Trends
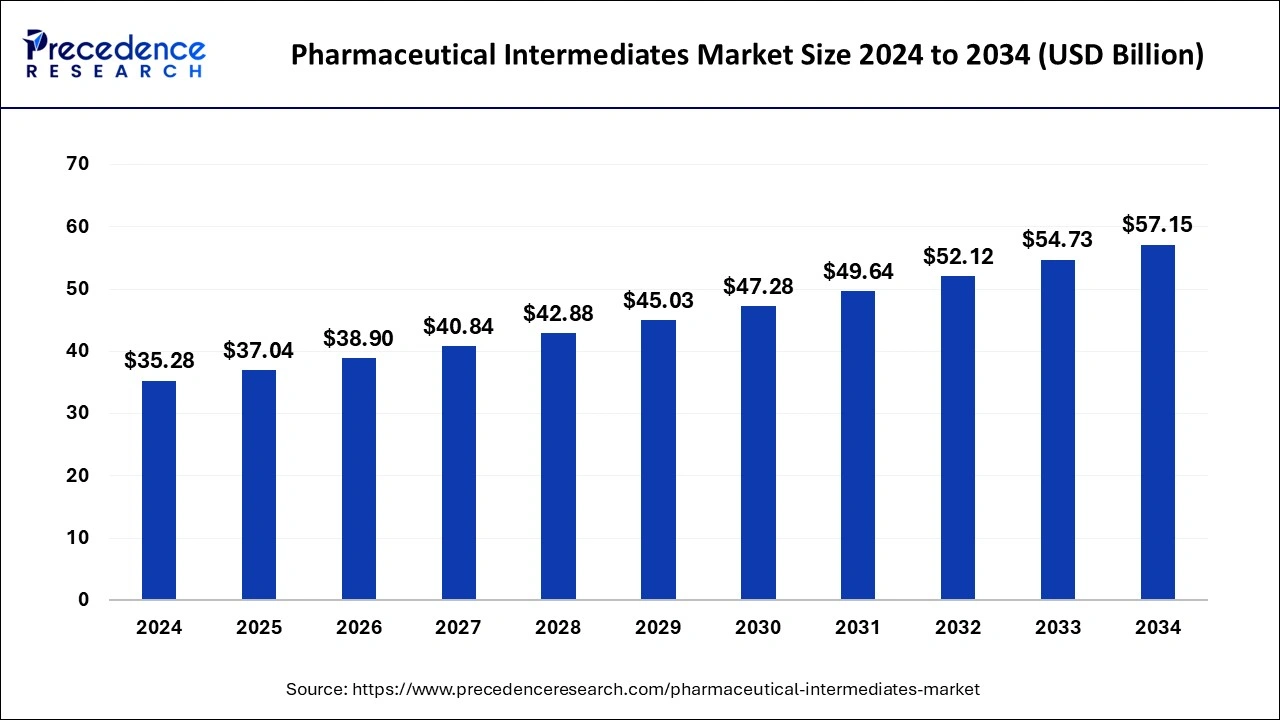
The global pharmaceutical intermediate market size is accounted at USD 37.04 billion in 2025 and is forecasted to hit around USD 57.15 billion by 2034, representing a CAGR of 4.94% from 2025 to 2034. (Source- Precedence Research)
This growth can be attributed to several factors, including the increasing demand for generic drugs, the rising prevalence of chronic diseases, and the expansion of pharmaceutical manufacturing activities in emerging markets.
The market growth is also driven by advancements in drug development and the need for efficient and cost-effective production processes. Pharmaceutical intermediates play a crucial role in the synthesis of active pharmaceutical ingredients (APIs) and the manufacturing of finished pharmaceutical products. These intermediates serve as building blocks and precursor materials, enabling the transformation of raw materials into complex molecules required for drug production.
Strengths
Weaknesses
Strengths
Robust Manufacturing Infrastructure: The pharmaceutical intermediate market benefits from well-established manufacturing facilities equipped with advanced technologies and efficient production processes. This infrastructure enables the production of high-quality intermediates in large quantities.
Strong Research and Development Capabilities: The industry’s focus on innovation and continuous improvement drives the development of novel synthesis routes and manufacturing processes. The ability to create and optimize efficient and cost-effective intermediate production methods is a significant strength
Regulatory Compliance: Pharmaceutical intermediates manufacturers are experienced in navigating complex regulatory frameworks and ensuring compliance with stringent quality and safety standards. Adhering to regulatory requirements enhances the credibility and trustworthiness of manufacturers.
Customization and Flexibility: Manufacturers often offer customized synthesis services to meet specific customer requirements. This ability to tailor intermediates to unique specifications provides a competitive edge and fosters strong customer relationships.
Weaknesses
Intellectual Property Risks: Developing proprietary synthesis routes and protecting intellectual property can be challenging. There is a risk of unauthorized duplication or infringement, which can undermine the competitive advantage of manufacturers.
Quality Control Challenges: Maintaining consistent quality across different batches of intermediates is a complex task. Ensuring batch-to-batch consistency, purity, and stability requires robust quality control systems and analytical testing protocols.
Dependence on Raw Materials: The availability and cost of raw materials can impact the production and profitability of pharmaceutical intermediates. Manufacturers need to manage the supply chain effectively to mitigate the risks associated with raw material sourcing.
Opportunities
Threats
Opportunities
Growing Pharmaceutical Industry: The global pharmaceutical industry is experiencing significant growth, driven by factors such as an ageing population, increased healthcare expenditure, and the rising prevalence of chronic diseases. This growth presents opportunities for the pharmaceutical intermediate market to expand its customer base and meet the growing demand for APIs and finished pharmaceutical products.
Increasing Outsourcing of Manufacturing: Pharmaceutical companies are increasingly outsourcing intermediate manufacturing to contract manufacturers to leverage cost advantages, access specialised expertise, and enhance operational flexibility. This trend creates opportunities for contract manufacturers to expand their customer base and develop long-term partnerships.
Emerging Markets: Emerging markets, particularly in Asia Pacific, Latin America, and the Middle East, offer significant growth potential for pharmaceutical intermediates. The expanding pharmaceutical industries in these regions, coupled with favourable government initiatives and rising healthcare investments, present opportunities for market expansion and collaboration.
Threats
Regulatory Challenges: Evolving regulatory requirements and increasing scrutiny from regulatory authorities pose challenges for manufacturers. Compliance with complex regulations, including GMP standards, can be resource-intensive and impact production timelines.
Competitive Landscape: The pharmaceutical intermediate market is highly competitive, with several established players and new entrants. Intense competition can lead to price pressures and reduced profit margins, especially when combined with factors such as raw material price fluctuations and market volatility.
Supply Chain Disruptions: Disruptions in the supply chain, such as natural disasters, trade conflicts, or pandemics, can impact the availability of raw materials and hinder production. Manufacturers need to implement effective supply chain management strategies to mitigate these risks.
Technological Advancements: While technological advancements provide opportunities, they also pose threats. Rapidly evolving technologies can render existing manufacturing processes obsolete, requiring manufacturers to continually invest in research and development to stay competitive.
APIs vs. Intermediates: Key Differences
FAQs-
1. What are pharmaceutical intermediates?
Pharmaceutical intermediates are chemical compounds used as building blocks during the multi-step synthesis of active pharmaceutical ingredients (APIs) or finished drug products.
2. Why are intermediates important in drug development?
They enable complex chemical transformations, support molecule stability, enhance bioavailability, and ensure the final drug meets required quality and regulatory standards.
3. What factors are driving growth in the pharmaceutical intermediates market?
Growth is driven by rising demand for generics, increasing chronic disease prevalence, expansion of manufacturing in emerging markets, and the need for efficient, cost-effective synthesis processes.
4. What challenges do manufacturers face when producing intermediates?
Challenges include maintaining quality consistency, managing raw material supply, protecting intellectual property, and meeting evolving regulatory requirements.
5. Why are pharmaceutical companies outsourcing intermediate manufacturing?
Companies outsource to access specialised synthesis expertise, reduce costs, improve flexibility, and scale production efficiently while focusing on core R&D.
6. What should procurement teams consider when partnering with intermediate manufacturers?
They should ensure clear communication, strong quality systems, well-defined roles, cost-management strategies, robust risk mitigation, and long-term relationship building.
Download our CDMO News Tracker to stay ahead of every shift in the CDMO landscape.
Download our CDMO News Tracker to stay ahead of every shift in the CDMO landscape.









