What is a CDMO?
A Contract Development and Manufacturing Organization (CDMO) is a specialized service provider that partners with pharmaceutical and biotechnology companies to handle both the development and the manufacturing of drug products — from early-stage research through to commercial-scale production. Unlike a traditional contract manufacturer, a CDMO integrates development expertise with GMP manufacturing infrastructure, offering a single partner capable of supporting a drug candidate from IND-enabling studies all the way through regulatory approval and market launch. For the thousands of pharma and biotech companies that lack the capital, capacity, or technical breadth to manufacture drugs in-house, CDMOs have become an indispensable strategic partner in bringing medicines to patients.
CDMO Market Size
The global pharmaceutical CDMO market size is valued at USD 197.40 billion in 2025 and is predicted to increase from USD 211 billion in 2026 to approximately USD 368.70 billion by 2034, expanding at a CAGR of 7.15% from 2025 to 2034 (Source- Precedence Research)
This explosive growth trajectory reflects a fundamental transformation in pharmaceutical manufacturing, where outsourcing has evolved from a cost-optimization strategy into a strategic imperative for accessing specialized expertise, cutting-edge technologies, and flexible production capacity. The CDMO sector now stands as the backbone of biopharmaceutical innovation, enabling companies to focus internal resources on drug discovery while leveraging external partners for development and manufacturing excellence.
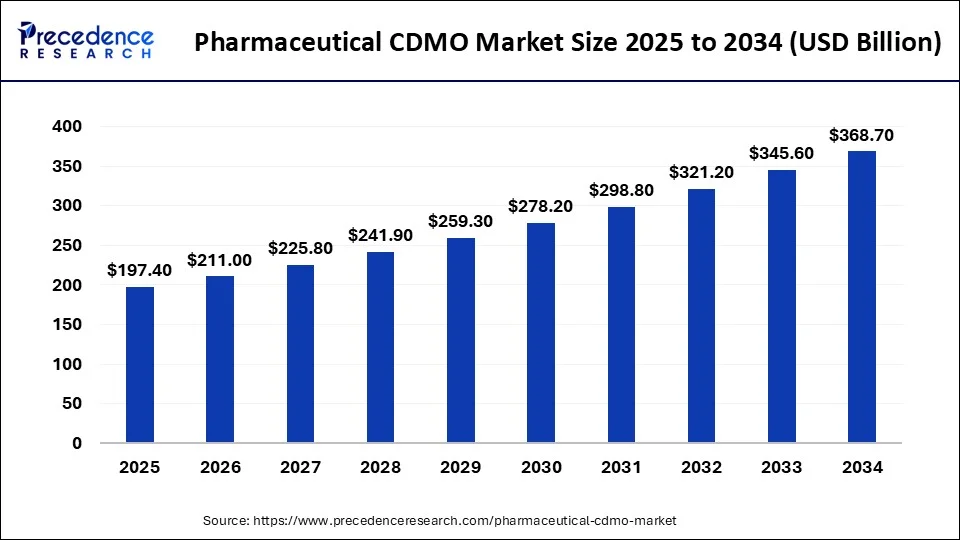
Four macro forces are reshaping the competitive and structural landscape of the global CDMO industry heading into the second half of this decade.
The first is the biologics and advanced therapy manufacturing wave. Monoclonal antibodies, gene therapies, cell therapies, and peptide therapeutics require capital-intensive, technically specialized manufacturing infrastructure that most sponsors cannot justify building in-house — driving sustained outsourcing demand that grows structurally with every new biologic approval.
The second is the GLP-1 peptide capacity surge. The extraordinary commercial success of semaglutide and tirzepatide has created acute fill-finish and peptide synthesis bottlenecks globally, pulling CDMO capacity toward this segment and reshaping the manufacturing strategies of sponsors across the industry.
The third is the ADC manufacturing boom. With over 300 antibody-drug conjugate candidates in clinical development as of 2025, CDMOs with high-potency API handling capabilities and conjugation platforms have become some of the most strategically valuable organizations in the sector — and one of the most capacity-constrained.
The fourth is M&A consolidation and the rise of end-to-end integrated CDMOs. Consolidation — typified by Novo Holdings’ $16.5 billion purchase of Catalent — signals a decisive shift toward end-to-end providers that combine development, scale-up, and commercial production. This trend is redrawing competitive dynamics and prompting sponsors to reassess whether specialist or integrated partners better serve their long-term manufacturing strategies.
What Does a CDMO Do?
A CDMO sits at the intersection of pharmaceutical science, engineering, regulatory expertise, and commercial manufacturing. Its core function is to accelerate a drug sponsor’s path from molecule to market by providing the development and production capabilities that most companies — particularly small and mid-sized biotechs — cannot economically build and sustain in-house.
CDMOs offer a range of services across the pharmaceutical development and manufacturing process, including:
-
API Development & Synthesis: Comprehensive small molecule and complex API development, route scouting, process chemistry, and scalable GMP manufacturing.
-
Drug Product Formulation Development: Pre-formulation studies, dosage form design, optimization, and technology transfer for solid, liquid, and injectable products.
-
Analytical Development & Validation: Method development, stability testing, impurity profiling, and full GMP validation to support regulatory filings.
-
Process Scale-Up & Optimization: Robust process development, tech transfer, and commercial-scale manufacturing readiness to ensure cost efficiency and reproducibility.
-
Clinical Supply Manufacturing: GMP production of clinical trial materials across Phase I, II, and III, including packaging, labeling, and global distribution support.
-
Sterile Fill-Finish Services: Aseptic processing, lyophilization, and injectable product manufacturing in compliance with global regulatory standards.
-
Secondary Packaging & Serialization: End-to-end packaging solutions, labeling, serialization, and supply chain compliance.
-
Regulatory Affairs & CMC Support: Strategic CMC documentation and submission support for IND, NDA, and BLA filings with the FDA, EMA, and other global regulatory authorities.
-
End-to-End Lifecycle Partnership: Engagement beginning at the IND-enabling stage through Phase I–III clinical development, regulatory approval, and commercial manufacturing, positioning the CDMO as a long-term strategic manufacturing partner.
The CDMO sector continues to grow and diversify, with an ever-increasing range of companies to choose from:
Essential CDMO Resources
To help with getting the most from the CDMO sector we have created a number of useful resources:
Quality Oversight Plan for CDMOs [Download]
Request For Quote (RFQ) Checklist [DOWNLOAD]
European CDMO Landscape Infographic
Top Technologies for European CDMOs
What is a CDMO, CMO and CRO?
In the pharmaceutical industry, there are several types of service providers that companies can partner with, each offering a different range of services. Three of these are Contract Development and Manufacturing Organizations (CDMOs), Contract Manufacturing Organizations (CMOs), and Contract Research Organizations(CROs).
Key differences between them include:
Contract Research Organizations (CRO) primarily focuses on providing research services. These services can include everything from early-stage research and development to clinical trials and regulatory affairs. By partnering with a CRO, pharmaceutical companies can access specialised expertise and infrastructure for their research needs.
Contract Manufacturing Organizations (CMO), on the other hand, specialises in manufacturing services. They provide the infrastructure and expertise needed to manufacture pharmaceutical products on a large scale. This allows pharmaceutical companies to bring their products to market without having to invest in their own manufacturing facilities.
Contract Development and Manufacturing Organizations (CDMO) provides a combination of both research and manufacturing services. They offer end-to-end support for pharmaceutical companies, from the early stages of research and development all the way through to manufacturing and packaging of the final product.
By understanding these differences, pharmaceutical companies can make informed decisions about which type of organisation to partner with based on their phase and specific needs.

Read CDMOs vs CROs vs CMOs: Understanding Key Services at Each Stage of Drug Development
The Role of a CDMO in Drug Development and Manufacturing
CDMOs can support pharmaceutical companies at every step of the way, from drug development to commercial manufacturing. This allows pharmaceutical companies to focus on their core competencies, such as drug discovery and marketing, while leaving the complex tasks of drug development and manufacturing to experts.
At the early stages of drug development, a CDMO can provide services such as pre-formulation and formulation development. Pre-formulation involves studying the physical and chemical properties of a drug, which is essential for designing effective and stable formulations. Formulation development then involves creating the actual drug product, which could be in various forms such as tablets, capsules, liquids, or injectables.
Once the drug formulation is developed, the CDMO can also assist with stability studies. These studies are critical for determining the shelf life of the drug product and understanding how different environmental factors might affect its stability.
As the drug moves into the clinical trial phase, a CDMO can provide clinical trial materials. These include the investigational medicinal product (IMP) that will be administered to patients during the trial, as well as the necessary documentation to ensure regulatory compliance.
Finally, once the drug is approved for marketing, a CDMO can scale up production from clinical trial quantities to commercial-scale quantities. They can also handle packaging and distribution of the final product.
Why Pharma companies use CDMOs
Pharmaceutical companies operate in a highly competitive and regulated environment. Developing a new drug is a complex, time-consuming, and expensive process. It involves numerous stages, each with its own set of challenges, from initial research and development to clinical trials and finally, manufacturing and marketing. This is where CDMOs come in.
CDMOs offer a range of benefits to pharmaceutical companies. Here are some key reasons why pharma companies choose to partner with CDMOs:
- Speed to Market: CDMOs have the expertise and resources to expedite the drug development process, helping pharmaceutical companies bring their products to market faster.
- Cost Savings: By outsourcing the drug development and manufacturing process to a CDMO, pharmaceutical companies can achieve significant cost savings. They can avoid the high capital expenditure associated with setting up their own manufacturing facilities and reduce operational costs.
- Scalability: CDMOs offer scalable solutions, allowing pharmaceutical companies to easily scale up or down their production based on demand.
- Focus on Core Competencies: By outsourcing the complex tasks of drug development and manufacturing to a CDMO, pharmaceutical companies can focus on their core competencies such as drug discovery and marketing.
- Regulatory Compliance: CDMOs are experienced in dealing with regulatory authorities around the world and can ensure that the drug development process complies with all relevant regulations.
- Access to Advanced Technologies: Many CDMOs have access to advanced technologies and specialised equipment that may not be available in-house for many pharmaceutical companies.

- Read our ranking of 552 FDF CDMO manufacturing sites across Europe, which reveals the countries with the most facilities in operation.
Types of CDMOs — Which Model Is Right for You?
The CDMO industry includes multiple business models, and selecting the right one is key to an effective outsourcing strategy.
Integrated / Full-Service CDMOs provide end-to-end capabilities from early development through commercial manufacturing across multiple modalities and geographies. They are ideal for sponsors seeking a single strategic partner with strong regulatory experience and robust quality systems.
Specialist CDMOs focus on specific technologies or modalities (e.g., viral vectors, HPAPIs, mRNA, sterile injectables). They offer deep technical expertise but have a narrower scope.
CRDMOs combine research, development, and manufacturing under one organization, supporting programs from discovery through commercialization while minimizing transition risks.
Dedicated vs. Multi-Client Facilities: Dedicated sites offer capacity certainty and exclusivity at a premium cost, while multi-client facilities provide cost efficiency and flexibility, especially for early-stage programs.
Virtual Pharma Demand: Asset-light biotech and pharma companies increasingly rely on CDMOs for manufacturing, making this the fastest-growing driver of outsourcing demand.
How to choose the right CDMO for your business
Selecting a CDMO is among the most consequential decisions a pharmaceutical or biotech company makes in its development journey.
A poor choice can cost years of delay and hundreds of millions of dollars. The following eight criteria should anchor every CDMO evaluation process.
1. Manufacturing modality fit. This is the single most important filter. A CDMO with deep experience in sterile injectables is not automatically a capable partner for viral vector manufacturing or ADC conjugation. Map your molecule’s specific chemistry, biology, and dosage form requirements against the CDMO’s documented platform capabilities before proceeding to any other evaluation.
2. GMP certification and regulatory inspection history. Request the CDMO’s complete inspection record from the FDA, EMA, MHRA, and any other relevant authority. Review all Form 483 observations and warning letters from the past five years, and require written evidence of completed CAPA. A clean, well-managed inspection history is the most reliable proxy for the maturity of a CDMO’s quality systems.
3. Capacity availability and expansion plans. Confirm actual available capacity for your program’s specific timeline — not theoretical maximum capacity. Ask how the CDMO manages priority conflicts between clients during capacity pressure, and whether any planned CapEx expansions are funded and on schedule.
4. Regulatory submission track record. If you expect the CDMO to support your NDA, BLA, or ANDA filing, verify they have done so successfully before. Request examples of CMC modules for submissions where they were the drug substance or drug product manufacturer of record. Confirm whether they maintain a Drug Master File with the FDA and EMA, and whether it is current.
5. In-house analytical and QC capabilities. Weak in-house analytical capability is one of the most common and expensive surprises in a CDMO relationship. Confirm the CDMO can execute the full suite of release and characterization testing for your molecule without relying on multiple external laboratories — which adds cost, timeline risk, and complexity to every batch release.
6. Technology transfer experience and on-time track record. Ask specifically about technology transfer timeline performance: what percentage of transfers are completed on schedule, and what are the most common causes of delay. Request references from sponsors who have transferred programs to — and ideally from — the CDMO.
7. Quality systems culture. The difference between a CDMO that passes inspections and one with genuine quality culture is significant and not always visible in the initial assessment. Look for low batch failure rates, rapid CAPA closure, low QA/QC staff turnover, and a site leadership team that treats quality as a business priority rather than a regulatory compliance exercise.
8. Commercial terms: IP, exclusivity, and pricing flexibility. Intellectual property ownership must be unambiguous from day one. Ensure the contract clearly defines ownership of process improvements, analytical methods, and manufacturing data generated during the relationship. Understand whether pricing is fixed-fee or cost-plus, and build in flexibility provisions for volume changes, clinical hold scenarios, and program termination.
In this video, David Caron SVP CMC at Ayala Pharmaceuticals explained his framework for selecting a Contract Manufacturing partner:

Market Dynamics
Growth Drivers
-
Rising Pharma Outsourcing – Companies focus on R&D while leveraging CDMOs to avoid $500M–$2B facility costs and long build timelines.
-
Increasing Drug Complexity – Biologics, CAR-T, HPAPIs, and ADCs require specialized tech and containment, driving CDMO reliance.
-
Growing Chronic Disease Burden – Rising cancer, diabetes, obesity, and autoimmune cases sustain long-term biologics demand.
-
Patent Cliffs & Biosimilars – Biologic LOEs fuel biosimilar growth, requiring advanced manufacturing expertise.
-
Technology Advancement – AI, continuous manufacturing, PAT, and single-use systems boost efficiency and flexibility.
-
GLP-1 Demand Surge: Explosive prescription growth driving >$100B sales forecasts by early 2030s; tightening fill-finish and peptide synthesis capacity
-
FDA Approval Momentum: 50 novel drug approvals in 2024, many advanced biologics, translating directly into long-term commercial CDMO manufacturing demand
Key Growth Inhibitors in the CDMO Market
-
Capacity Constraints: Ongoing bottlenecks in viral vectors, HPAPI, and aseptic fill-finish infrastructure
-
Talent Shortages: Limited GMP-skilled workforce across North America and Europe, intensified by GLP-1 manufacturing demand
-
Geopolitical Risk: US biosecurity legislation and China-linked CDMO concerns accelerating supply chain diversification
-
Rising Regulatory Scrutiny: Increasing compliance pressure, particularly for complex biologics and gene-editing therapies
-
Elevated Risk Profile: Greater reliance on specialist partners increases both strategic value and regulatory exposure
Tune into the PharmaSource Podcast for exclusive CDMO insights from leading industry experts.
Tune into the PharmaSource Podcast for exclusive CDMO insights from leading industry experts.
Frequently Asked Questions About CDMOs
What does CDMO stand for?
CDMO stands for Contract Development and Manufacturing Organization. It is a company that provides pharmaceutical and biotechnology clients with both drug development services — including process development, formulation, and analytical work — and GMP-compliant manufacturing services, from clinical trial supply through commercial-scale production.
What is the difference between a CDMO, CMO, and CRO?
A CMO (Contract Manufacturing Organization) provides production services only — it manufactures a drug product to the sponsor’s existing specification without contributing development expertise. A CDMO adds development capabilities to that manufacturing relationship, offering integrated service from process design through scaled production. A CRO (Contract Research Organization) provides clinical research services — trial management, data analysis, and regulatory strategy — but does not manufacture drug products. The CRDMO model merges research, development, and manufacturing into a single integrated service platform.
Why do pharmaceutical companies use CDMOs?
The primary reasons are access to specialized manufacturing capabilities, capital efficiency, speed to market, and regulatory expertise. Building a GMP-compliant biologics manufacturing facility can cost $500 million or more and take five to seven years. Partnering with a CDMO eliminates that capital burden and accelerates clinical and commercial timelines significantly. For virtual and asset-light biotech companies, CDMOs are the entire manufacturing infrastructure — not simply a supplement to in-house capability.
How do I choose the right CDMO for my drug?
Start with manufacturing modality fit — the CDMO must have documented experience with your specific drug type, dosage form, and manufacturing process. Then evaluate GMP certification and inspection history, capacity availability, regulatory submission track record, in-house analytical capability, technology transfer performance, quality systems maturity, and commercial terms including IP ownership and pricing flexibility. PharmaSource provides a searchable database of CDMOs filterable by all of these criteria.
What is an integrated or full-service CDMO?
An integrated or full-service CDMO offers end-to-end services across the entire drug development and manufacturing lifecycle — from IND-enabling studies, process and formulation development, and clinical trial supply through to commercial manufacturing, packaging, and post-approval support. The advantage for sponsors is reduced handoffs, consolidated regulatory accountability, and a partner with complete institutional knowledge of the molecule throughout its development history.
What GMP certifications should a CDMO have?
At minimum, a CDMO should hold valid manufacturing authorizations from the FDA (21 CFR Part 211 for finished drug products, 21 CFR Part 210 for APIs) and the EMA (EU GMP directives under EudraLex Volume 4) for every market in which you intend to commercialize your product. For biologics, compliance with ICH Q5–Q11 guidelines is essential. For clinical-stage manufacturing, EU GMP Annex 13 governs IMP production. For cell and gene therapies, 21 CFR Part 1271 and EudraLex Annex 2 apply. The CDMO’s inspection history with these agencies — specifically the number, severity, and resolution of observations — is a more meaningful quality indicator than the certifications themselves.
What is a CRDMO?
A CRDMO — Contract Research, Development and Manufacturing Organization — is an integrated outsourcing model combining research services (typically preclinical biology, medicinal chemistry, or discovery chemistry) with development and manufacturing capabilities. The model is designed for sponsors who want a single partner from early discovery through commercial manufacturing, minimizing the knowledge transitions and technology transfer risks that arise when programs move between separate research and production organizations.



















